-
Calcium ions (Ca2+) play a key role of the nerve cells generating universal intracellular signals and controlling important functions. Ca2+ activation is of great significance for explaining the subcellular-level biological process. Light stimulated nerve cells to control intracellular signals and membrane activities has become a main method in neuroscience, and the photoactivation is one of the main ways to study intracellular Ca2+ transmission. Nerve cells can be directly stimulated by light to produce action potentials, but such techniques are inaccurate in the delivered light energy. To improve this, here in this work we show that gold nanorods (GNRs) can be conjugated to ligands to bound to human neuroblast cells (SH-SY5Y), and introduce an optical method of stimulating and monitoring Ca2+ signal in nerve cells in which the plasmonic excitation of GNRs is used. In this paper, we use confocal microscopy to display the 488 nm continuous wave laser irradiating SH-SY5Y cells with Ca2+ indicator (Fluo-4, AM) to check fluorescence. Near-infrared pulsed light at the plasmon resonance absorption peak of GNRs is used to stimulate Ca2+ signal transduction in SH-SY5Y labeled with GNRs, and Fluo-4, AM is used for two-photon excited fluorescence imaging. In addition, we use the pulsed laser with power of 0.5 mW and a wavelength of 800 nm. The Ca2+ activation can be achieved in 10 s on average. The release rate of Ca2+ from SH-SY5Y cells labeled with GNRs is 6 times that without GNRs. Next, in order to determine the source of changes in Ca2+, we use the BPATA to deplete the intracellular Ca2+, after 5 min, 200 μmol/L Ca2+ solution is added, and its ΔF/F is found to be more than 1.5 times that without GNRs. Thus, we believe that GNRs could enhance photoactivation through local surface plasmon resonance induced membrane depolarization and generate an action potential. The results prove the feasibility of using GNRs to enhance the activation of Ca2+ in nerve cells, and provide an optical means of lower photodamage and more precise for studying nerve cell ion channels. Our study demonstrates that enhancing photoactivation by GNRs could provide an outlook of basic research in neuroscience.
-
Keywords:
- nerve cell /
- calcium ion /
- photoactivation /
- two-photon excited fluorescence
[1] Michael J H, Bernardo L S 2008 Neuron 59 902
 Google Scholar
Google Scholar
[2] Richard D F, Dong H W, Peter J B 2015 Neuron 86 374
 Google Scholar
Google Scholar
[3] John C W, Emilia E 2015 Biophys. J. 108 1934
 Google Scholar
Google Scholar
[4] Camilo R, Mariateresa T, Paolo M, Attilio M, Gianni C, Sergio M, Roberto R 2018 J. Neural Eng. 15 036016
 Google Scholar
Google Scholar
[5] Jean D, Luis H, Katrien M, Dries B, Dimiter P 2017 Front. Neurosci. 11 663
 Google Scholar
Google Scholar
[6] Lu C B, Wu X L, Ma H Z, Wang Q C, Wang Y K, Luo Y, Li C, Xu H 2019 Neural Plast . 2019 5271573
 Google Scholar
Google Scholar
[7] Robert K S, Joel V, Owen B, David A X N 2018 J. Neural Eng. 15 041004
 Google Scholar
Google Scholar
[8] Jacob G B, Edward S B 2011 Trends Cogn. Sci. 15 592
 Google Scholar
Google Scholar
[9] Yao J P, Hou W S, Yin Z Q 2012 Int J Ophthalmol. 5 517
 Google Scholar
Google Scholar
[10] Sonny G, Terence S L, Clare E E, Ilias T 2014 Biomed. Opt. Express 5 2896
 Google Scholar
Google Scholar
[11] Flavie L C, Charleen S, Éric B, Michel M, Paul D K 2016 Sci. Rep. 6 1
 Google Scholar
Google Scholar
[12] Paviolo C, Thompson A C, Yong J, Brown W G, Stoddart P R 2014 J. Neural Eng. 11 065002
 Google Scholar
Google Scholar
[13] de Boer W.D.A.M, Hirtz J J, Capretti A, Gregorkiewicz T, Izquierdo-Serra M, Han S, Dupre C, Shymkiv Y, Yuste R 2018 Light Sci. Appl. 7 100
 Google Scholar
Google Scholar
[14] Shapiro M G, Homma K, Villarreal S, Richter C P, Bezanilla F 2012 Nat. Commun. 3 736
 Google Scholar
Google Scholar
[15] João L C D S, Jeremy S T, Bobo D, Stephen B H K, David R P, Francisco B 2015 Neuron 86 207
 Google Scholar
Google Scholar
[16] Morven C, Orsolya K, John W M, Jonathan T, Paul P B, André V S, Yossi B 2016 PLoS One 11 e0155468
 Google Scholar
Google Scholar
[17] Silvia S, Aniello S M, Vito D M 2014 Gen. Physiol. Biophys. 33 121
 Google Scholar
Google Scholar
[18] Shen B L, Yan J S, Wang S Q, Zhou F F, Zhao Y H, Hu R, Qu J L, Liu L W 2020 Theranostics 10 1849
 Google Scholar
Google Scholar
[19] Lin F R, Pintu D, Zhao Y H, Shen B L, Hu R, Zhou F F, Liu L W, Qu J L 2020 Biomed. Opt. Express 11 149
 Google Scholar
Google Scholar
[20] Kyungsik E, Kyung M B, Sang B J, Sung J K, Jonghwan L 2018 Biophys. J. 115 1481
 Google Scholar
Google Scholar
[21] João L C D S, Okhil K N, Eunkeu Oh, Alan L H, Igor V, David R P, Francisco B, James B, Delehanty 2019 ACS Chem. Neurosci. 10 1478
 Google Scholar
Google Scholar
[22] Michael B R, Chad A M, George C S 2016 J. Phys. Chem. C 120 816
 Google Scholar
Google Scholar
[23] Vincenzo A, Roberto P, Marco F, Onofrio M M, Maria A I 2017 J. Phys. Condens. Matter 29 203002
 Google Scholar
Google Scholar
[24] Ni W H, Kou X S, Yang Z, Wang J F 2008 ACS Nano 2 677
 Google Scholar
Google Scholar
[25] Sassaroli E, Li K C P, O’Neill B E 2009 Phys. Med. Biol. 54 5541
 Google Scholar
Google Scholar
[26] Ekici O, Harrison R K, Durr N J, Eversole D S, Lee M, Yakar A B 2008 J. Phys. D 41 185501
 Google Scholar
Google Scholar
[27] Luo L Q 2015 Principles of Neurobiology (New York: Garland Science) pp40−43
[28] Roberta D A, Pasquale P, Giuseppe S 2017 Beilstein J. Nanotechnol. 8 1
 Google Scholar
Google Scholar
-
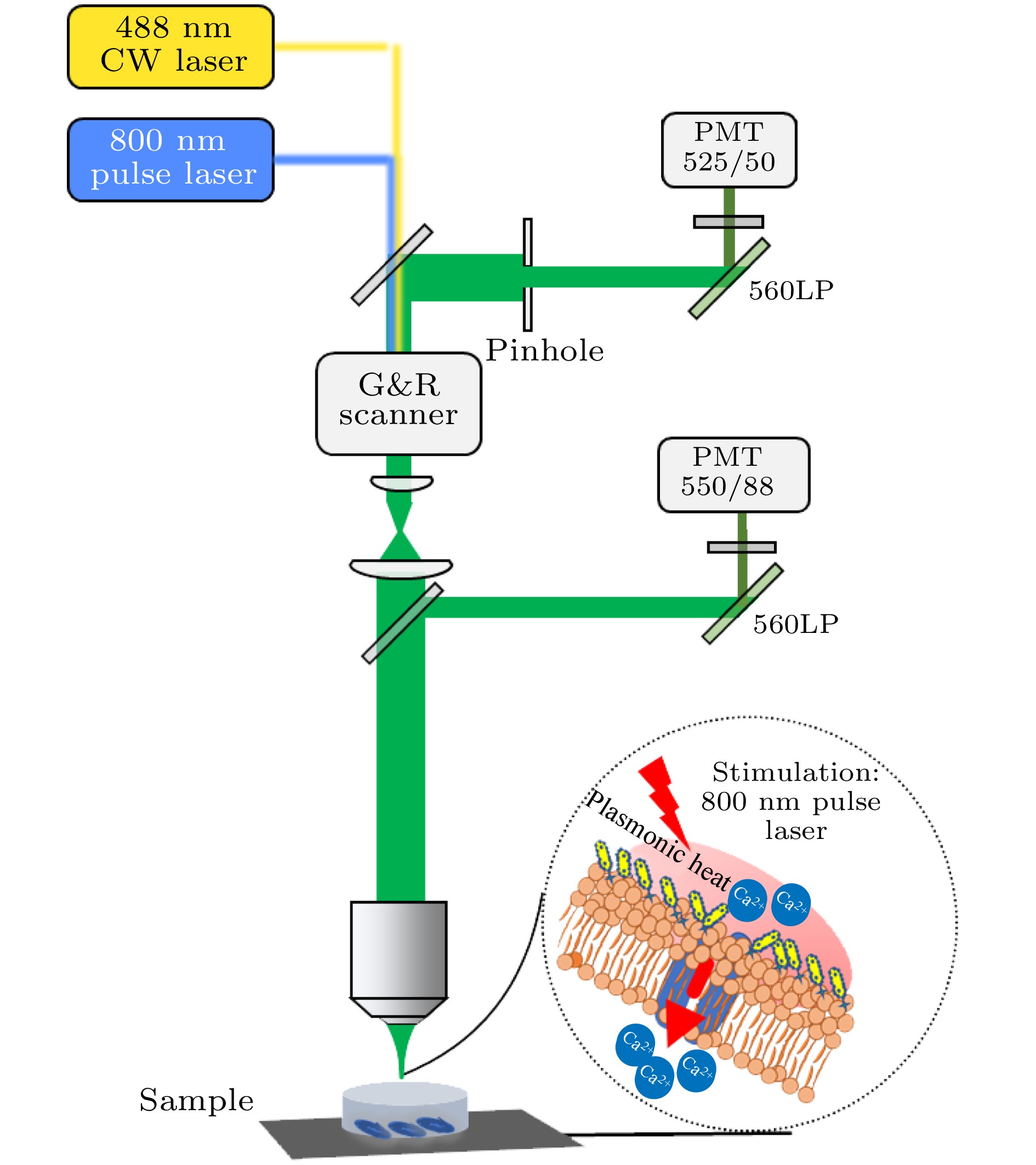
图 1 双光子激发荧光共聚焦显微系统光路示意图(G&R Scanner为振镜扫描仪, LP为长通滤波片, PMT为光电倍增管)
Figure 1. Schematic diagram of two-photon excited fluorescence confocal microscopy system. The abbreviations in the figure are as follows: G&R Scanner is a Galvo-resonance scanner, LP is a long-pass filter, and PMT is a photomultiplier tube.
图 2 温度随时间t和距离x变化的函数 (a)时间一定时, 温度随着距离的增加而衰减; (b) 距离一定时, 温度随着时间的增加而衰减; 在此模型中, 选用520 W·cm–2, 间隔为0.5 ms的矩形脉冲作为激发光源
Figure 2. Temperature as a function of time t and distance x: (a) When the distance is constant, the temperature decays with increasing time; (b) when the time is constant, the temperature decays with increasing distance. In this model, a rectangular pulse of 520 W·cm–2 with an interval of 0.5 ms is selected as the excitation light source.
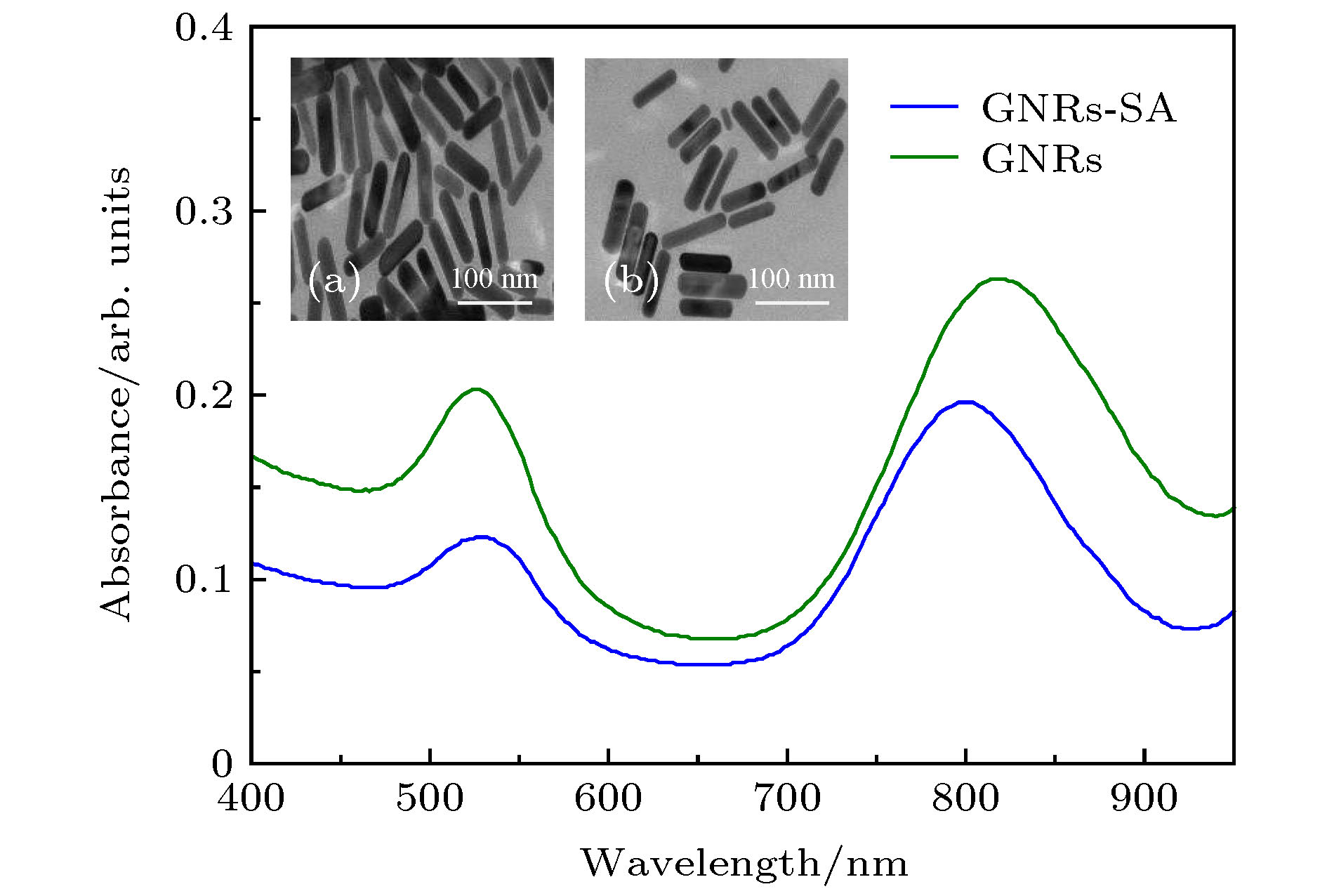
图 3 GNRs与GNRs-SA的(a) 可见近红外吸收光谱以及(b) TEM图(比例尺: 100 nm; 近红外区GNRs\GNRs-SA的吸收峰分别在820和800 nm)
Figure 3. (a) Visible near-infrared absorption spectra and (b) TEM images of GNRs and GNRs-SA, scale bar: 100 nm. the absorption peaks of GNRs\GNRs-SA in the near infrared region were at 820 nm and 800 nm, respectively.
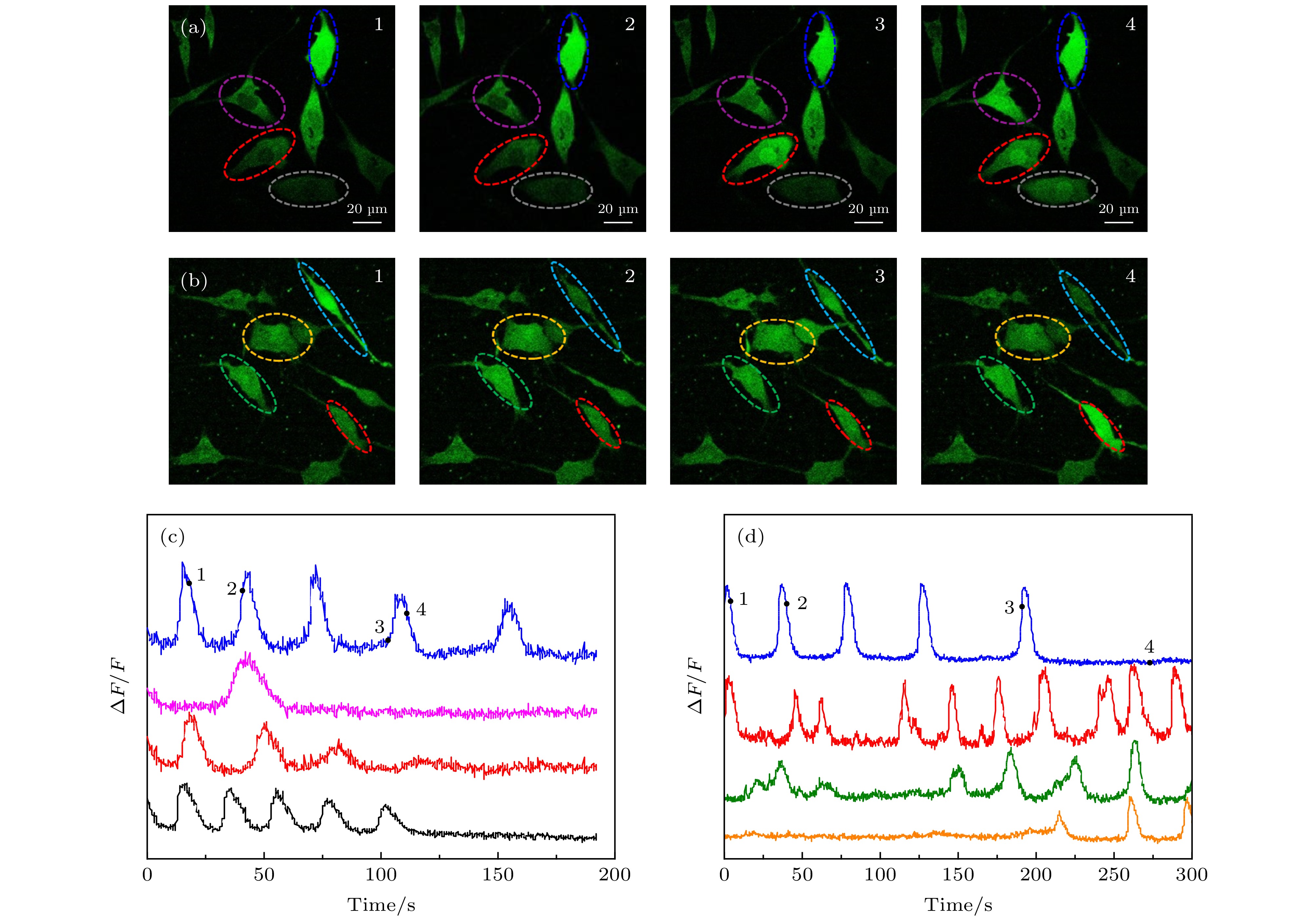
图 4 仅Fluo-4, AM标记与Fluo-4, AM, Con A和GNRs-SA同时标记的SH-SY5Y细胞光激活成像 (a) Fluo-4, AM, Con A和GNRs-SA标记的SH-SY5Y不同时刻的荧光图像; (b) Fluo-4, AM标记的SH-SY5Y不同时刻的荧光图像; (c) Fluo-4, AM, Con A和GNRs-SA标记的SH-SY5Y相对荧光强度随时间变化的曲线, 数字“1, 2, 3, 4”跟(a)中图像的时间点(25 s, 54 s, 99 s, 116 s)相对应, 四条曲线对应(a)中不同颜色圈出的细胞; (d) Fluo-4, AM标记的SH-SY5Y相对荧光强度随时间变化的曲线, 数字“1, 2, 3, 4”跟(b)中图像的时间点(1 s, 32 s, 127 s, 187 s)相对应, 四条曲线对应(b)中不同颜色圈出的细胞
Figure 4. Photoactivation SH-SY5Y cells imaging of only Fluo-4 labeled and labeled with Fluo-4, AM, Con A and GNRs-SA. (a) The fluorescence images of Fluo-4, AM and GNRs-SA labeled SH-SY5Y cells at different times. (b) the fluorescence images of Fluo-4, AM labeled SH-SY5Y cells at different times. (c) the changing of relative fluorescence intensity of Fluo-4, AM, Con A and GNRs-SA labeled SH-SY5Y cells with time. the numbers “1, 2, 3, 4” correspond to the time points (25 s, 54 s, 99 s, 116 s) of the image in (a), and the four curves correspond to the cells circled in different colors in (a). (d) The changing of relative fluorescence intensity of Fluo-4, AM labeled SH-SY5Y cells with time. the numbers “1, 2, 3, 4” correspond to the time points (1 s, 32 s, 127 s, 187 s) of the image in (b). four curves correspond to cells circled in different colors in (b).
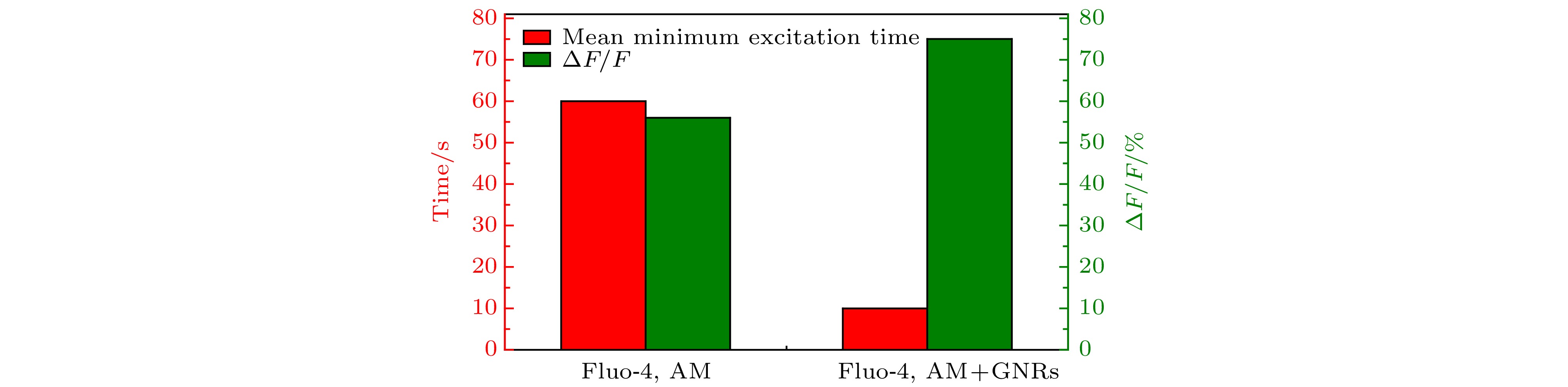
图 5 仅Fluo-4, AM标记和Fluo-4, AM, Con A和GNRs-SA同时标记的SH-SY5Y细胞光刺激的平均(细胞数n = 300)最小激发时间以及光激活引起相对荧光强度的变化值
Figure 5. For Fluo-4, AM labeled and Fluo-4, AM, Con A and GNRs-SA labeled SH-SY5Y cells at the same time, the average photoactivation (cell number n = 300) minimum excitation time and the changes in relative fluorescence intensity caused by photoactivation.
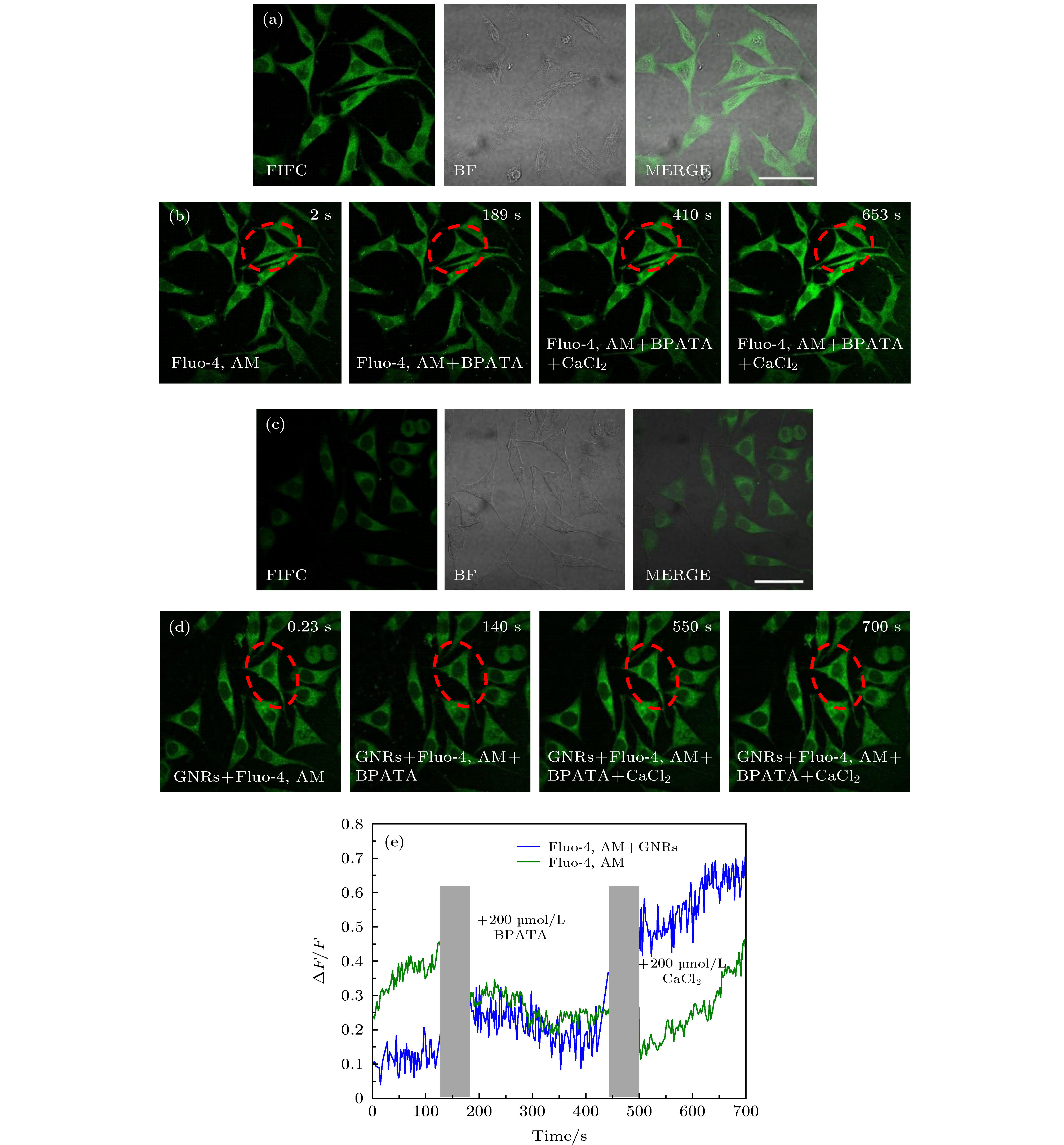
图 6 验证光激活时Ca2+的变化来源(其中FITC表示使用500—550 nm的带通滤波片来采集荧光, BF表示透射成像, Merge表示两通道的叠加图) (a) Fluo-4, AM染色后SH-SY5Y细胞的荧光图像和透射光图像 (比例尺: 50 μm); (b) Fluo-4, AM染色后SH-SY5Y细胞不同时刻的双光子荧光图像; (c) Fluo-4, AM与2 µmol/L Con A, GNRs-SA孵育后SH-SY5Y细胞的荧光图像和透射光图像 (比例尺: 50 μm); (d) Fluo-4, AM与2 µmol/L Con A, GNRs-SA 孵育后SH-SY5Y细胞不同时刻的双光子荧光图像; (e) 图(b)和(d)中红色虚线所圈区域的ΔF/F在相继添加200 µmol/L BPATA与200 µmol/L CaCl2后的变化曲线
Figure 6. Verify the changing of the source of Ca2 + during photoactivation. Among them, FITC indicates the use of a 500—550 nm band-pass filter to collect fluorescence, BF indicates transmission imaging, and Merge indicates an overlay of two channels. (a) Fluorescence and TD image of SH-SY5Y cells with Fluo-4, AM staining (scale bar: 50 μm). (b) Two-photon fluorescence images of SH-SY5Y cells labeled with Fluo-4, AM at different times. (c) Fluorescence and TD images of SH-SY5Y cells with Fluo-4, AM staining after adding 2 µmol/L Con A, GNRs-SA (scale bar: 50 μm). (d) Time-series two-photon fluorescence images of SH-SY5Y cells labeled with Fluo-4, AM staining after adding 2 µmol/L Con A, GNRs-SA. (e) The change curves of the ΔF/F of the red virtual circle areas in panel (b) and (d) after adding 200 µmol/L BPATA and 200 µmol/L CaCl2.
-
[1] Michael J H, Bernardo L S 2008 Neuron 59 902
 Google Scholar
Google Scholar
[2] Richard D F, Dong H W, Peter J B 2015 Neuron 86 374
 Google Scholar
Google Scholar
[3] John C W, Emilia E 2015 Biophys. J. 108 1934
 Google Scholar
Google Scholar
[4] Camilo R, Mariateresa T, Paolo M, Attilio M, Gianni C, Sergio M, Roberto R 2018 J. Neural Eng. 15 036016
 Google Scholar
Google Scholar
[5] Jean D, Luis H, Katrien M, Dries B, Dimiter P 2017 Front. Neurosci. 11 663
 Google Scholar
Google Scholar
[6] Lu C B, Wu X L, Ma H Z, Wang Q C, Wang Y K, Luo Y, Li C, Xu H 2019 Neural Plast . 2019 5271573
 Google Scholar
Google Scholar
[7] Robert K S, Joel V, Owen B, David A X N 2018 J. Neural Eng. 15 041004
 Google Scholar
Google Scholar
[8] Jacob G B, Edward S B 2011 Trends Cogn. Sci. 15 592
 Google Scholar
Google Scholar
[9] Yao J P, Hou W S, Yin Z Q 2012 Int J Ophthalmol. 5 517
 Google Scholar
Google Scholar
[10] Sonny G, Terence S L, Clare E E, Ilias T 2014 Biomed. Opt. Express 5 2896
 Google Scholar
Google Scholar
[11] Flavie L C, Charleen S, Éric B, Michel M, Paul D K 2016 Sci. Rep. 6 1
 Google Scholar
Google Scholar
[12] Paviolo C, Thompson A C, Yong J, Brown W G, Stoddart P R 2014 J. Neural Eng. 11 065002
 Google Scholar
Google Scholar
[13] de Boer W.D.A.M, Hirtz J J, Capretti A, Gregorkiewicz T, Izquierdo-Serra M, Han S, Dupre C, Shymkiv Y, Yuste R 2018 Light Sci. Appl. 7 100
 Google Scholar
Google Scholar
[14] Shapiro M G, Homma K, Villarreal S, Richter C P, Bezanilla F 2012 Nat. Commun. 3 736
 Google Scholar
Google Scholar
[15] João L C D S, Jeremy S T, Bobo D, Stephen B H K, David R P, Francisco B 2015 Neuron 86 207
 Google Scholar
Google Scholar
[16] Morven C, Orsolya K, John W M, Jonathan T, Paul P B, André V S, Yossi B 2016 PLoS One 11 e0155468
 Google Scholar
Google Scholar
[17] Silvia S, Aniello S M, Vito D M 2014 Gen. Physiol. Biophys. 33 121
 Google Scholar
Google Scholar
[18] Shen B L, Yan J S, Wang S Q, Zhou F F, Zhao Y H, Hu R, Qu J L, Liu L W 2020 Theranostics 10 1849
 Google Scholar
Google Scholar
[19] Lin F R, Pintu D, Zhao Y H, Shen B L, Hu R, Zhou F F, Liu L W, Qu J L 2020 Biomed. Opt. Express 11 149
 Google Scholar
Google Scholar
[20] Kyungsik E, Kyung M B, Sang B J, Sung J K, Jonghwan L 2018 Biophys. J. 115 1481
 Google Scholar
Google Scholar
[21] João L C D S, Okhil K N, Eunkeu Oh, Alan L H, Igor V, David R P, Francisco B, James B, Delehanty 2019 ACS Chem. Neurosci. 10 1478
 Google Scholar
Google Scholar
[22] Michael B R, Chad A M, George C S 2016 J. Phys. Chem. C 120 816
 Google Scholar
Google Scholar
[23] Vincenzo A, Roberto P, Marco F, Onofrio M M, Maria A I 2017 J. Phys. Condens. Matter 29 203002
 Google Scholar
Google Scholar
[24] Ni W H, Kou X S, Yang Z, Wang J F 2008 ACS Nano 2 677
 Google Scholar
Google Scholar
[25] Sassaroli E, Li K C P, O’Neill B E 2009 Phys. Med. Biol. 54 5541
 Google Scholar
Google Scholar
[26] Ekici O, Harrison R K, Durr N J, Eversole D S, Lee M, Yakar A B 2008 J. Phys. D 41 185501
 Google Scholar
Google Scholar
[27] Luo L Q 2015 Principles of Neurobiology (New York: Garland Science) pp40−43
[28] Roberta D A, Pasquale P, Giuseppe S 2017 Beilstein J. Nanotechnol. 8 1
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 15040
- PDF Downloads: 168
- Cited By: 0














 DownLoad:
DownLoad: