-
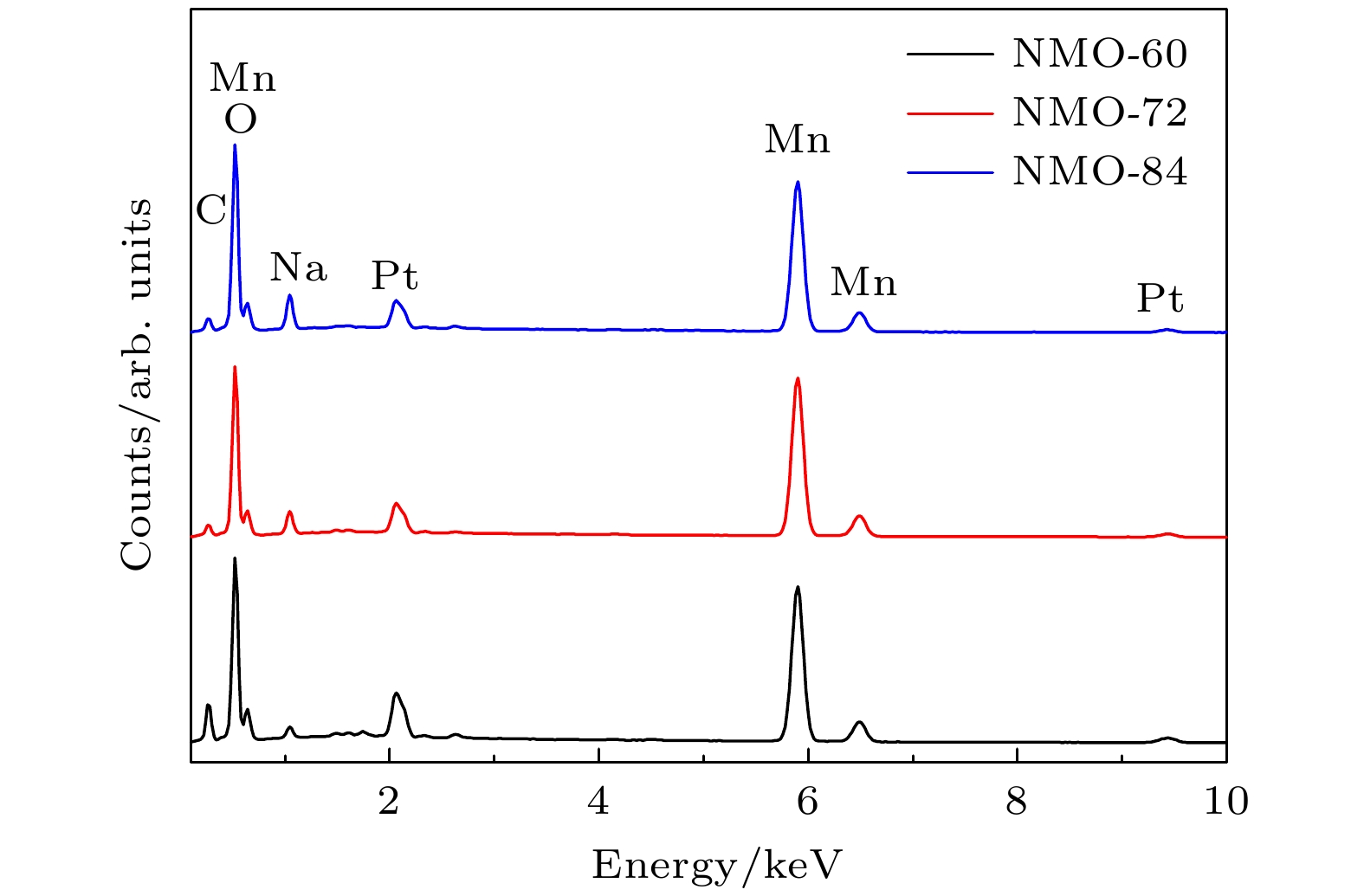
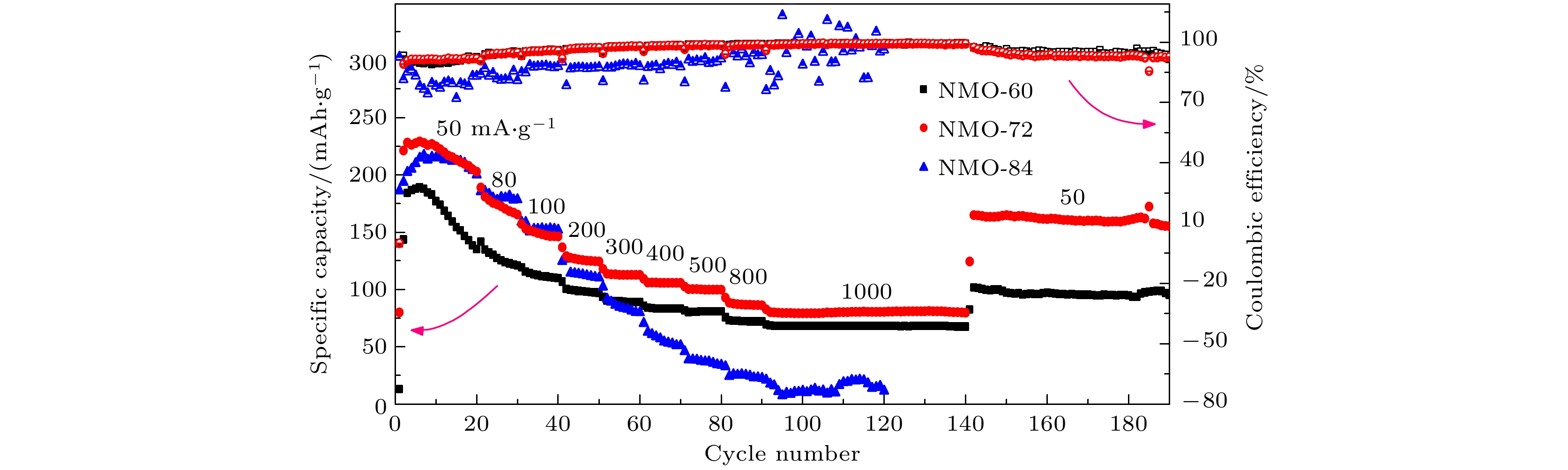
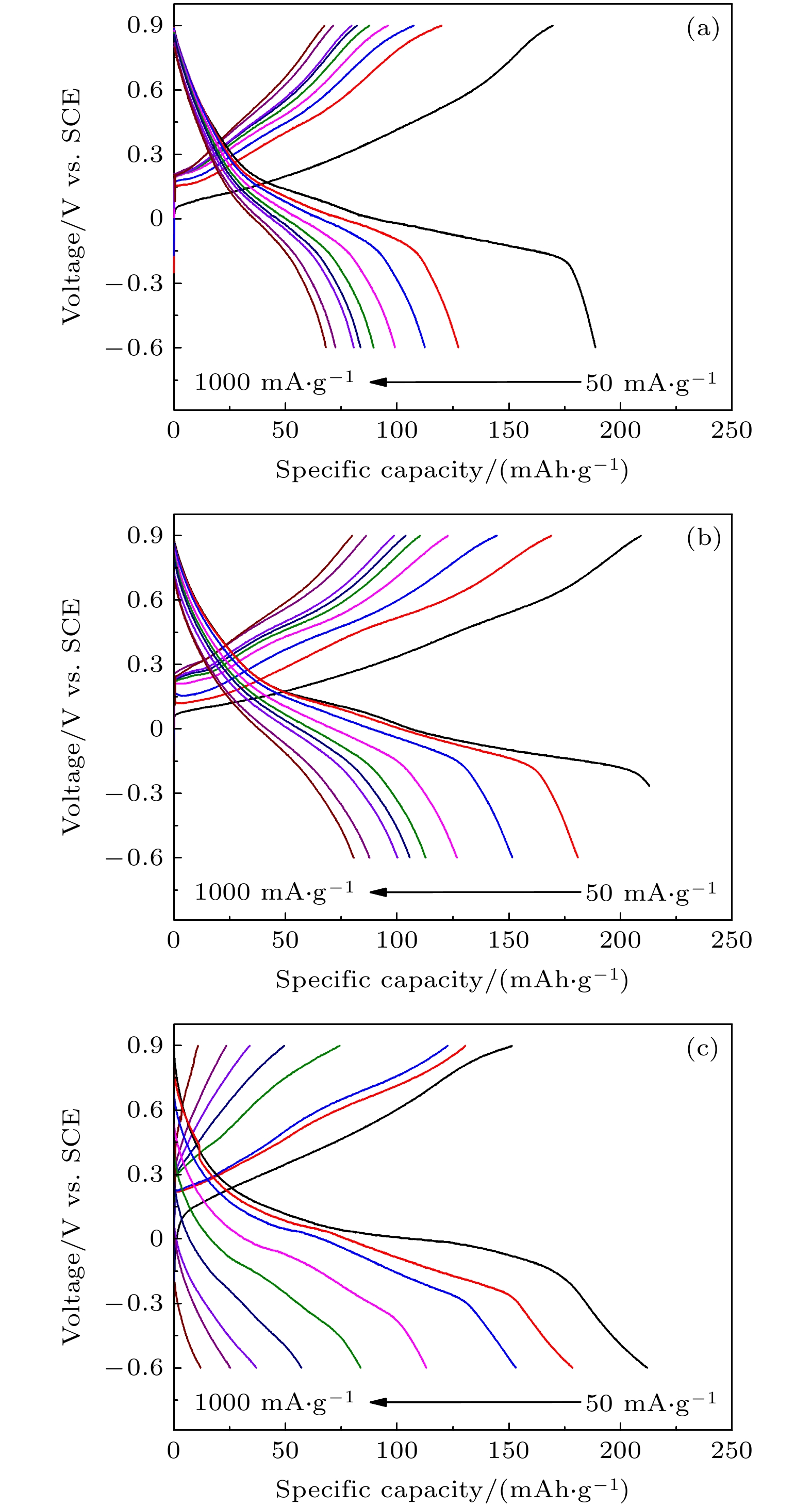
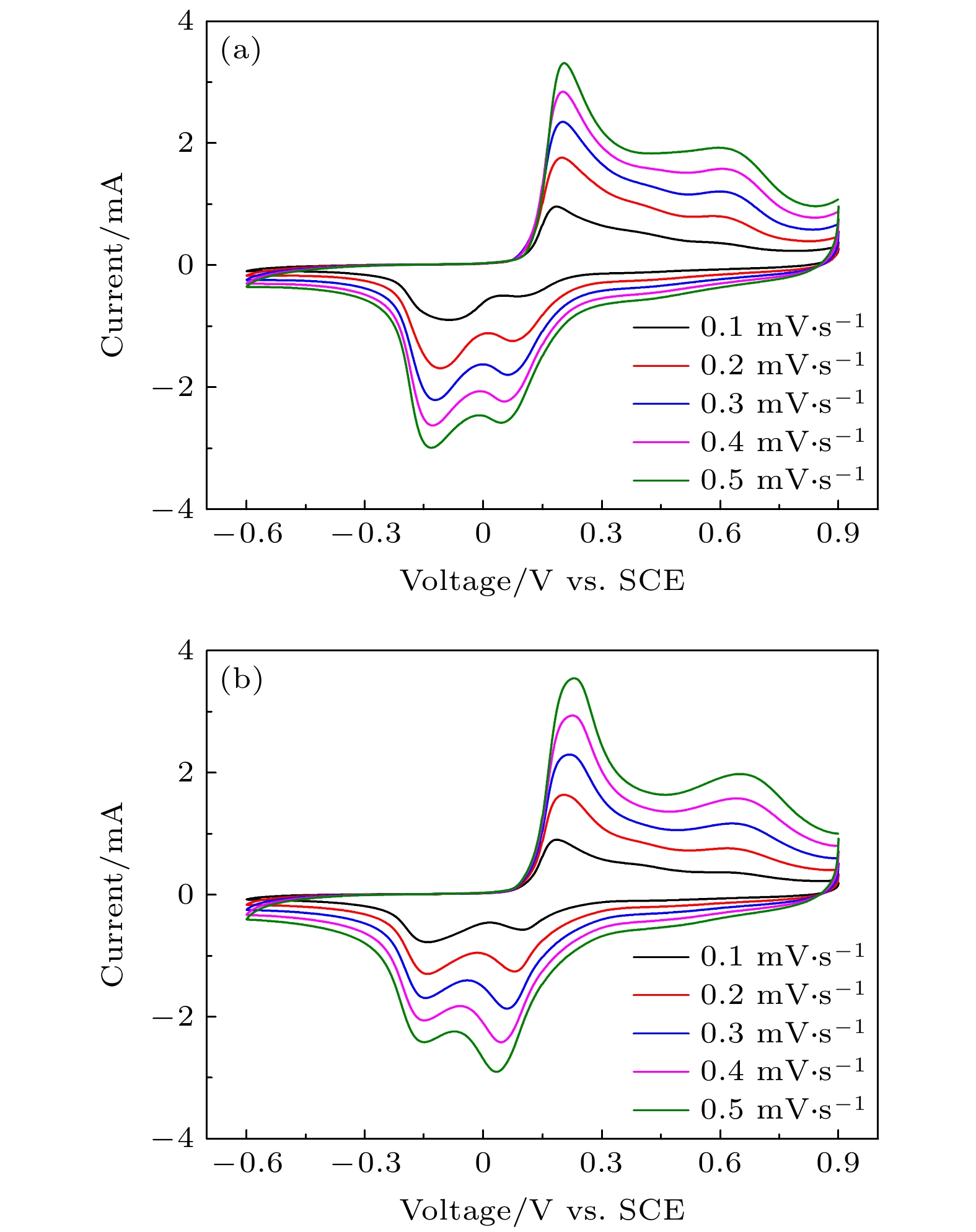
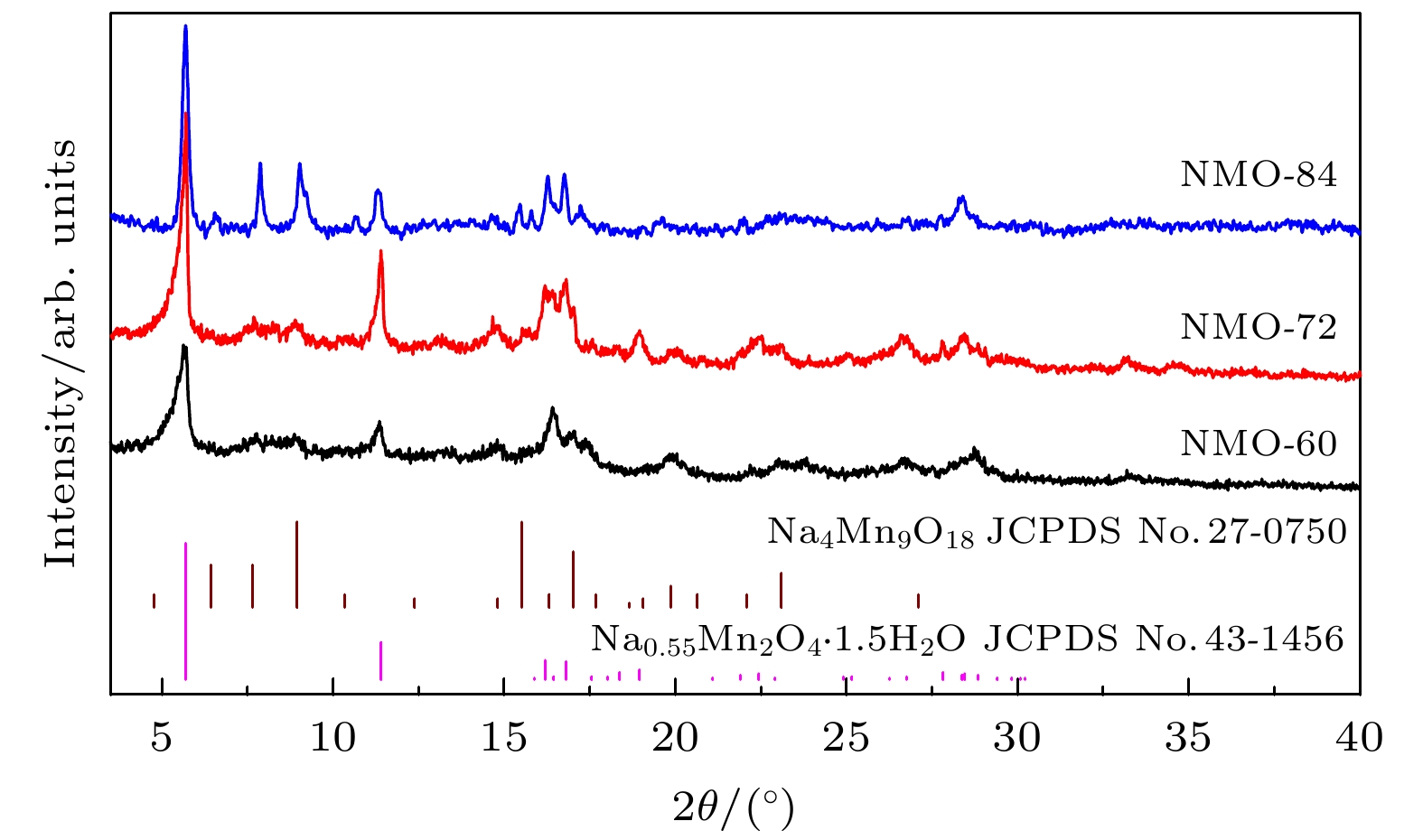
近年来镁离子电池正极材料的研发成为研究热点, 提高电池的高倍率性能和循环稳定性成为主要研究目标. 本文采用水热法制备了纳米线和纳米片共混结构的钠锰氧化物(Na0.55Mn2O4·1.5H2O)纳米材料, 并用X−射线衍射和扫描电子显微镜进行表征, 通过充放电测试仪和电化学工作站进行变倍率充放电循环和变扫速循环伏安测试. 结果表明水热反应时间对材料的相结构和形貌组成影响显著, 其中水热反应72 h样品(NMO-72)中纳米片和纳米线形成紧密共混, 纳米片有效填充到纳米线交叉空隙中, 提高了材料的振实密度. 并且NMO-72材料具有更高的放电比容量和倍率循环性能. 在50 mA·g–1电流密度下, NMO-72的放电比容量达到229.1 mAh·g–1; 在1000 mA·g–1电流密度下, NMO-72材料的放电比容量稳定在81 mAh·g–1; 而电流密度再次回到50 mA·g–1时, 其放电比容量稳定保持在164.7 mAh·g–1. 同时, 循环伏安测试表明NMO-72材料与其他材料相比, 具有最佳的镁离子扩散动力学性能, 由此NMO-72材料具有更为优异的可逆比容量、高倍率性能和循环稳定性.In recent years, the cathode materials of magnesium ion batteries have become a hot point of research, and the improvement of high-rate performance and cycle stability has become the main research goal. In this paper, sodium manganese oxide (Na0.55Mn2O4·1.5H2O) nanomaterial with a blended structure of nanowires and nanosheets is prepared by the hydrothermal method. The structure and morphology of the material are analyzed by X-ray diffraction and scanning electron microscopy. The variable rate charge-discharge curves and variable scan rate cyclic voltammetry curves are obtained by a battery tester and electrochemical workstation, respectively. The results show that the hydrothermal reaction time has significant effects on phase structure and morphology composition of the material. The nanosheets and nanowires in the sample form a closely blend by 72-h hydrothermal reaction (NMO-72), and the nanosheets effectively fill into the intersecting space of the nanowires. In this way, the tap density of the material is improved. More importantly, NMO-72 has higher discharge specific capacity and rate cycling performance. At a current density of 50 mA·g–1, the discharge specific capacity of NMO-72 reaches 229.1 mAh·g–1. At a current density of 1000 mA·g–1, the discharge specific capacity of the NMO-72 stabilizes at 81 mAh·g–1. When the current density returns to 50 mA·g–1 again, the discharge specific capacity remains stable at 164.7 mAh·g–1. Besides, the cyclic voltammetry test shows that the NMO-72 material has more excellent magnesium ion diffusion kinetic performance than other materials. Therefore, the NMO-72 material has more excellent reversible specific capacity, high rate performance and cycling stability.
-
Keywords:
- aqueous Mg-ion batteries /
- cathode materials /
- sodium manganese oxides /
- electrochemical performance
[1] Muldoon J, Bucur C B, Gregory T 2014 Chem. Rev. 114 11683
 Google Scholar
Google Scholar
[2] Huie M M, Bock D C, Takeuchi E S, Marschilok A C, Takeuchi K J 2015 Coord. Chem. Rev. 287 15
 Google Scholar
Google Scholar
[3] 刘文龙, 黄可贤, 周学俊, 李驰麟 2020 硅酸盐学报 48 978
 Google Scholar
Google Scholar
Liu W L, Huang K X, Zhou X J, Li C L 2020 J. Chin. Ceram. Soc. 48 978
 Google Scholar
Google Scholar
[4] 刘凡凡, 王田甜, 范丽珍 2020 硅酸盐学报 48 947
 Google Scholar
Google Scholar
Liu F F, Wang T T, Fan L Z 2020 J. Chin. Ceram. Soc. 48 947
 Google Scholar
Google Scholar
[5] 苏硕剑, 努丽燕娜, 非路热·吐尔逊, 杨军, 王久林 2015 物理化学学报 31 111
 Google Scholar
Google Scholar
Su S J, Nuli Y N, Feilure T, Yang J, Wang J L 2015 Acta Phys. Chim. Sin. 31 111
 Google Scholar
Google Scholar
[6] Song J, Sahadeo E, Noked M, Lee S B 2016 J. Phys. Chem. Lett. 7 1736
 Google Scholar
Google Scholar
[7] Xu M, Lei S, Qi J, Dou Q, Liu L, Lu Y, Huang Q, Shi S, Yan X 2018 ACS Nano 12 3733
 Google Scholar
Google Scholar
[8] Xu C, Chen Y, Shi S, Li J, Kang F, Su D 2015 Sci. Rep. 5 14120
 Google Scholar
Google Scholar
[9] Zhang Y, Liu G, Zhang C, Chi Q, Zhang T, Feng Y, Zhu K, Zhang Y, Chen Q, Cao D 2020 Chem. Eng. J. 392 123652
 Google Scholar
Google Scholar
[10] Pan H, Shao Y, Yan P, Cheng Y, Han K S, Nie Z, Wang C, Yang J, Li X, Bhattacharya P, Mueller K T, Liu J 2016 Nat. Energy 1 16039
 Google Scholar
Google Scholar
[11] Liang Y, Jing Y, Gheytani S, Lee K Y, Liu P, Facchetti A, Yao Y 2017 Nat. Mater. 16 841
 Google Scholar
Google Scholar
[12] Zhang H, Ye K, Zhu K, Cang R, Wang X, Wang G, Cao D 2017 ACS Sustainable Chem. Eng. 5 6727
 Google Scholar
Google Scholar
[13] Arthur T S, Zhang R, Ling C, Glans P A, Fan X, Guo J, Mizuno F 2014 ACS Appl. Mater. Interfaces 6 7004
 Google Scholar
Google Scholar
[14] Liu G, Chi Q, Zhang Y, Chen Q, Zhang C, Zhu K, Cao D 2018 Chem. Commun. 54 9474
 Google Scholar
Google Scholar
[15] Liu M, Jain A, Rong Z, Qu X, Canepa P, Malik R, Ceder G, Persson K A 2016 Energy Environ. Sci. 9 3201
 Google Scholar
Google Scholar
[16] Saha P, Jampani P H, Datta M K, Hong D, Gattu B, Patel P, Kadakia K S, Manivannan A, Kumta P N 2017 Nano Res. 10 4415
 Google Scholar
Google Scholar
[17] NuLi Y, Yang J, Li Y, Wang J 2010 Chem. Commun. 46 3794
 Google Scholar
Google Scholar
[18] 李卓, 宁哲, 刘坤, 王一雍, 韩露, 路金林 2017 中国冶金 27 1
 Google Scholar
Google Scholar
Li Z, Ning Z, Liu K, Wang Y Y, Han L, Lu J L 2017 China Metall. 27 1
 Google Scholar
Google Scholar
[19] 李艳阳, 熊跃, 张建民, 陈卫华 2015 材料导报 29 50
 Google Scholar
Google Scholar
Li Y Y, Xiong Y, Zhang J M, Chen W H 2015 Mater. Rep. 29 50
 Google Scholar
Google Scholar
[20] Zhang J, He T, Zhang W, Sheng J Z, Amiinu I S, Kou Z K, Yang J L, Mai L Q, Mu S C 2017 Adv. Energy Mater. 7 1602092
 Google Scholar
Google Scholar
[21] 杨顺毅, 王先友, 魏建良, 李秀琴, 唐安平 2008 物理化学学报 24 1669
 Google Scholar
Google Scholar
Yang S Y, Wang X Y, Wie J L, Li X Q, Tang A P 2008 Acta Phys. Chim. Sin. 24 1669
 Google Scholar
Google Scholar
[22] Xu M W, Niu Y B, Li Y T, Bao S J, Li C M 2014 RSC Adv. 4 30340
 Google Scholar
Google Scholar
[23] Lv W J, Huang Z G, Yin Y X, Yao H R, Zhu H L, Guo Y G 2019 ChemNanoMat 5 1253
 Google Scholar
Google Scholar
[24] Zheng P, Su J X, Wang Y B, Zhou W, Song J J, Su Q M, Reeves-McLaren N, Guo S W 2020 ChemSusChem 13 1793
 Google Scholar
Google Scholar
[25] 陆雅翔, 赵成龙, 容晓晖, 陈立泉, 胡勇胜 2018 67 120601
 Google Scholar
Google Scholar
Lu Y X, Zhao C L, Rong X H, Chen L Q, Hu Y S 2018 Acta Phys. Sin. 67 120601
 Google Scholar
Google Scholar
[26] Zhang Q N, Levi M D, Dou Q Y, Lu Y L, Chai Y G, Lei S L, Ji H X, Liu B, Bu X D, Ma P J, Yan X B 2019 Adv. Energy Mater. 9 1802707
 Google Scholar
Google Scholar
[27] Li J N, Yu J, Amiinu I S, Zhang J, Sheng J Z, Kou Z K, Wang Z, Yu Q, Mai L Q, Mu S C 2017 J. Mater. Chem. A 5 18509
 Google Scholar
Google Scholar
[28] Chua R, Cai Y, Kou Z K, Satish R, Ren H, Chan J J, Zhang L P, Morris S A, Bai J M, Srinivasan M 2019 Chem. Eng. J. 370 742
 Google Scholar
Google Scholar
[29] Wang X Y, Qin X H, Lu Q Q, Han M M, Omar A, Mikhailova D 2020 Chin. J. Chem. Eng. 28 2214
 Google Scholar
Google Scholar
[30] Xiao Y, Zhu Y F, Xiang W, Wu Z G, Li Y C, Lai J, Li S, Wang E, Yang Z G, Xu C L, Zhong B H, Guo X D 2020 Angew. Chem. Int. Ed. 59 1491
 Google Scholar
Google Scholar
[31] Okamoto S, Ichitsubo T, Kawaguchi T, Kumagai Y, Oba F, Yagi S, Shimokawa K, Goto N, Doi T, Matsubara E 2015 Angew. Adv. Sci. 2 1500072
 Google Scholar
Google Scholar
-
-
[1] Muldoon J, Bucur C B, Gregory T 2014 Chem. Rev. 114 11683
 Google Scholar
Google Scholar
[2] Huie M M, Bock D C, Takeuchi E S, Marschilok A C, Takeuchi K J 2015 Coord. Chem. Rev. 287 15
 Google Scholar
Google Scholar
[3] 刘文龙, 黄可贤, 周学俊, 李驰麟 2020 硅酸盐学报 48 978
 Google Scholar
Google Scholar
Liu W L, Huang K X, Zhou X J, Li C L 2020 J. Chin. Ceram. Soc. 48 978
 Google Scholar
Google Scholar
[4] 刘凡凡, 王田甜, 范丽珍 2020 硅酸盐学报 48 947
 Google Scholar
Google Scholar
Liu F F, Wang T T, Fan L Z 2020 J. Chin. Ceram. Soc. 48 947
 Google Scholar
Google Scholar
[5] 苏硕剑, 努丽燕娜, 非路热·吐尔逊, 杨军, 王久林 2015 物理化学学报 31 111
 Google Scholar
Google Scholar
Su S J, Nuli Y N, Feilure T, Yang J, Wang J L 2015 Acta Phys. Chim. Sin. 31 111
 Google Scholar
Google Scholar
[6] Song J, Sahadeo E, Noked M, Lee S B 2016 J. Phys. Chem. Lett. 7 1736
 Google Scholar
Google Scholar
[7] Xu M, Lei S, Qi J, Dou Q, Liu L, Lu Y, Huang Q, Shi S, Yan X 2018 ACS Nano 12 3733
 Google Scholar
Google Scholar
[8] Xu C, Chen Y, Shi S, Li J, Kang F, Su D 2015 Sci. Rep. 5 14120
 Google Scholar
Google Scholar
[9] Zhang Y, Liu G, Zhang C, Chi Q, Zhang T, Feng Y, Zhu K, Zhang Y, Chen Q, Cao D 2020 Chem. Eng. J. 392 123652
 Google Scholar
Google Scholar
[10] Pan H, Shao Y, Yan P, Cheng Y, Han K S, Nie Z, Wang C, Yang J, Li X, Bhattacharya P, Mueller K T, Liu J 2016 Nat. Energy 1 16039
 Google Scholar
Google Scholar
[11] Liang Y, Jing Y, Gheytani S, Lee K Y, Liu P, Facchetti A, Yao Y 2017 Nat. Mater. 16 841
 Google Scholar
Google Scholar
[12] Zhang H, Ye K, Zhu K, Cang R, Wang X, Wang G, Cao D 2017 ACS Sustainable Chem. Eng. 5 6727
 Google Scholar
Google Scholar
[13] Arthur T S, Zhang R, Ling C, Glans P A, Fan X, Guo J, Mizuno F 2014 ACS Appl. Mater. Interfaces 6 7004
 Google Scholar
Google Scholar
[14] Liu G, Chi Q, Zhang Y, Chen Q, Zhang C, Zhu K, Cao D 2018 Chem. Commun. 54 9474
 Google Scholar
Google Scholar
[15] Liu M, Jain A, Rong Z, Qu X, Canepa P, Malik R, Ceder G, Persson K A 2016 Energy Environ. Sci. 9 3201
 Google Scholar
Google Scholar
[16] Saha P, Jampani P H, Datta M K, Hong D, Gattu B, Patel P, Kadakia K S, Manivannan A, Kumta P N 2017 Nano Res. 10 4415
 Google Scholar
Google Scholar
[17] NuLi Y, Yang J, Li Y, Wang J 2010 Chem. Commun. 46 3794
 Google Scholar
Google Scholar
[18] 李卓, 宁哲, 刘坤, 王一雍, 韩露, 路金林 2017 中国冶金 27 1
 Google Scholar
Google Scholar
Li Z, Ning Z, Liu K, Wang Y Y, Han L, Lu J L 2017 China Metall. 27 1
 Google Scholar
Google Scholar
[19] 李艳阳, 熊跃, 张建民, 陈卫华 2015 材料导报 29 50
 Google Scholar
Google Scholar
Li Y Y, Xiong Y, Zhang J M, Chen W H 2015 Mater. Rep. 29 50
 Google Scholar
Google Scholar
[20] Zhang J, He T, Zhang W, Sheng J Z, Amiinu I S, Kou Z K, Yang J L, Mai L Q, Mu S C 2017 Adv. Energy Mater. 7 1602092
 Google Scholar
Google Scholar
[21] 杨顺毅, 王先友, 魏建良, 李秀琴, 唐安平 2008 物理化学学报 24 1669
 Google Scholar
Google Scholar
Yang S Y, Wang X Y, Wie J L, Li X Q, Tang A P 2008 Acta Phys. Chim. Sin. 24 1669
 Google Scholar
Google Scholar
[22] Xu M W, Niu Y B, Li Y T, Bao S J, Li C M 2014 RSC Adv. 4 30340
 Google Scholar
Google Scholar
[23] Lv W J, Huang Z G, Yin Y X, Yao H R, Zhu H L, Guo Y G 2019 ChemNanoMat 5 1253
 Google Scholar
Google Scholar
[24] Zheng P, Su J X, Wang Y B, Zhou W, Song J J, Su Q M, Reeves-McLaren N, Guo S W 2020 ChemSusChem 13 1793
 Google Scholar
Google Scholar
[25] 陆雅翔, 赵成龙, 容晓晖, 陈立泉, 胡勇胜 2018 67 120601
 Google Scholar
Google Scholar
Lu Y X, Zhao C L, Rong X H, Chen L Q, Hu Y S 2018 Acta Phys. Sin. 67 120601
 Google Scholar
Google Scholar
[26] Zhang Q N, Levi M D, Dou Q Y, Lu Y L, Chai Y G, Lei S L, Ji H X, Liu B, Bu X D, Ma P J, Yan X B 2019 Adv. Energy Mater. 9 1802707
 Google Scholar
Google Scholar
[27] Li J N, Yu J, Amiinu I S, Zhang J, Sheng J Z, Kou Z K, Wang Z, Yu Q, Mai L Q, Mu S C 2017 J. Mater. Chem. A 5 18509
 Google Scholar
Google Scholar
[28] Chua R, Cai Y, Kou Z K, Satish R, Ren H, Chan J J, Zhang L P, Morris S A, Bai J M, Srinivasan M 2019 Chem. Eng. J. 370 742
 Google Scholar
Google Scholar
[29] Wang X Y, Qin X H, Lu Q Q, Han M M, Omar A, Mikhailova D 2020 Chin. J. Chem. Eng. 28 2214
 Google Scholar
Google Scholar
[30] Xiao Y, Zhu Y F, Xiang W, Wu Z G, Li Y C, Lai J, Li S, Wang E, Yang Z G, Xu C L, Zhong B H, Guo X D 2020 Angew. Chem. Int. Ed. 59 1491
 Google Scholar
Google Scholar
[31] Okamoto S, Ichitsubo T, Kawaguchi T, Kumagai Y, Oba F, Yagi S, Shimokawa K, Goto N, Doi T, Matsubara E 2015 Angew. Adv. Sci. 2 1500072
 Google Scholar
Google Scholar
计量
- 文章访问数: 9874
- PDF下载量: 190
- 被引次数: 0













 下载:
下载: