-
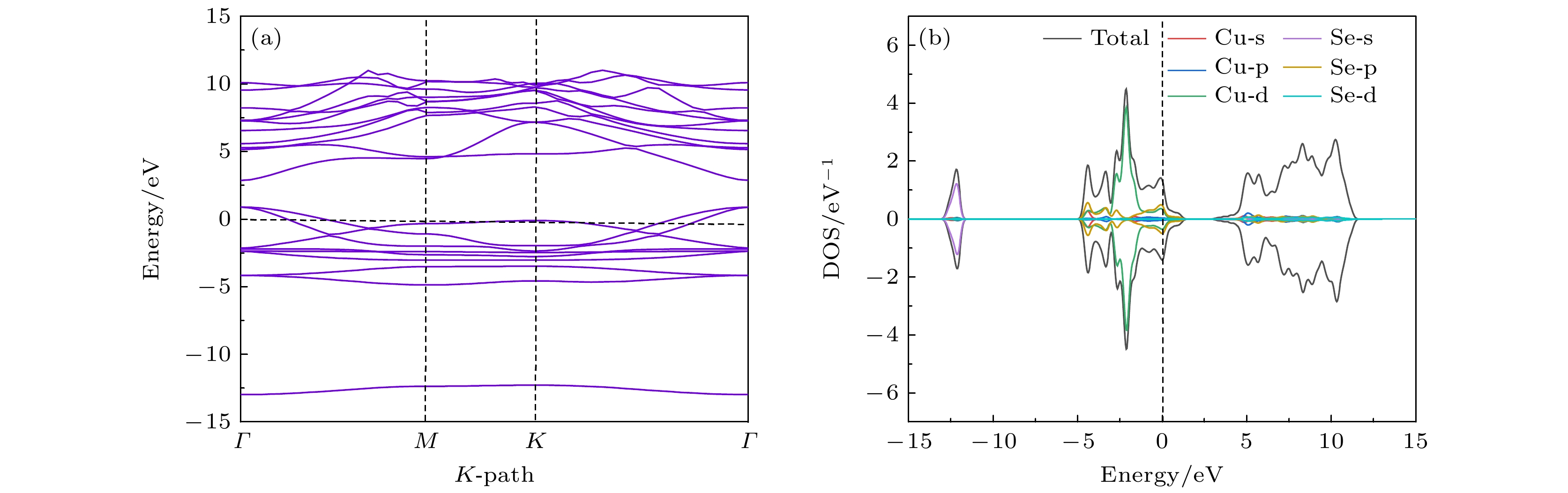
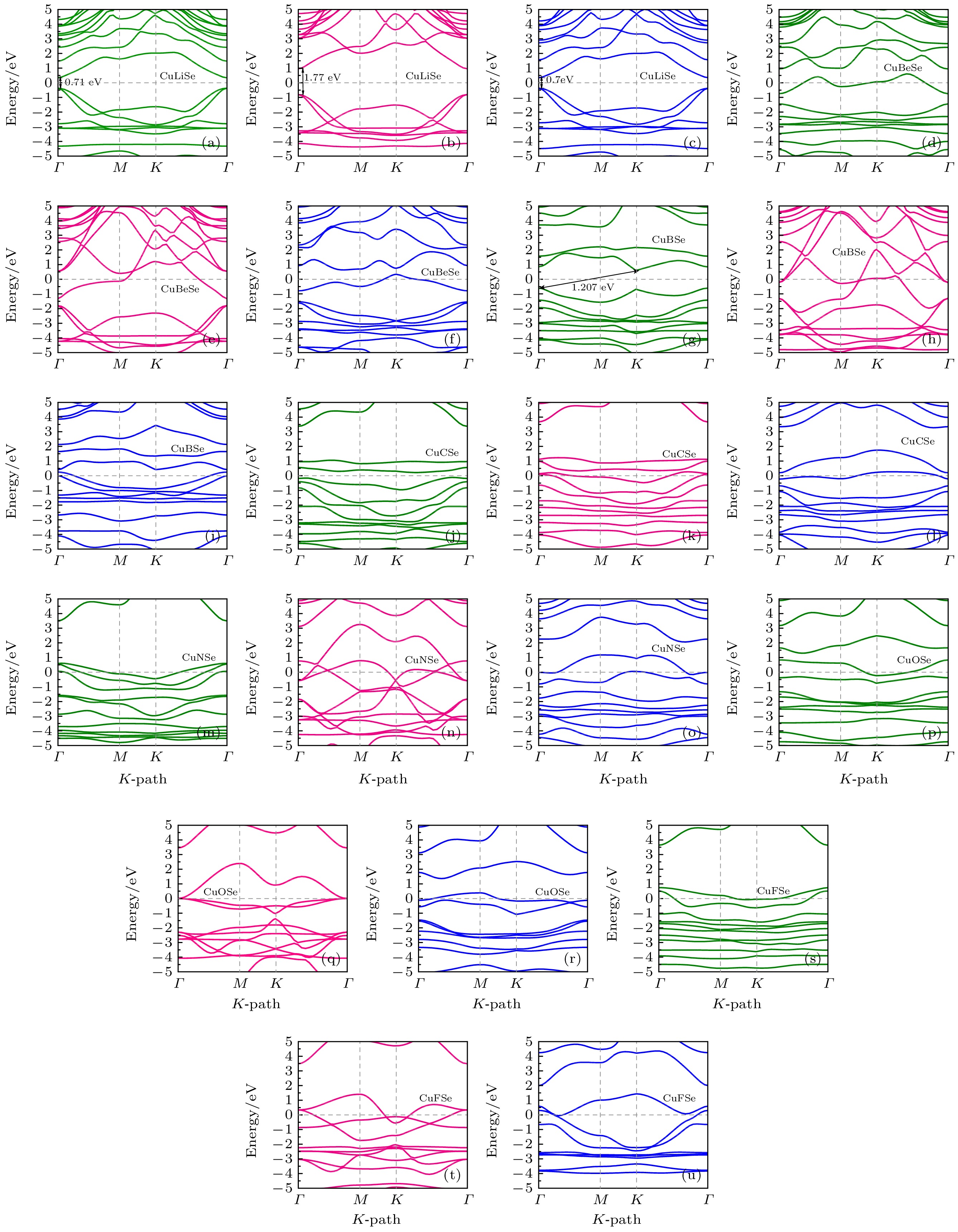
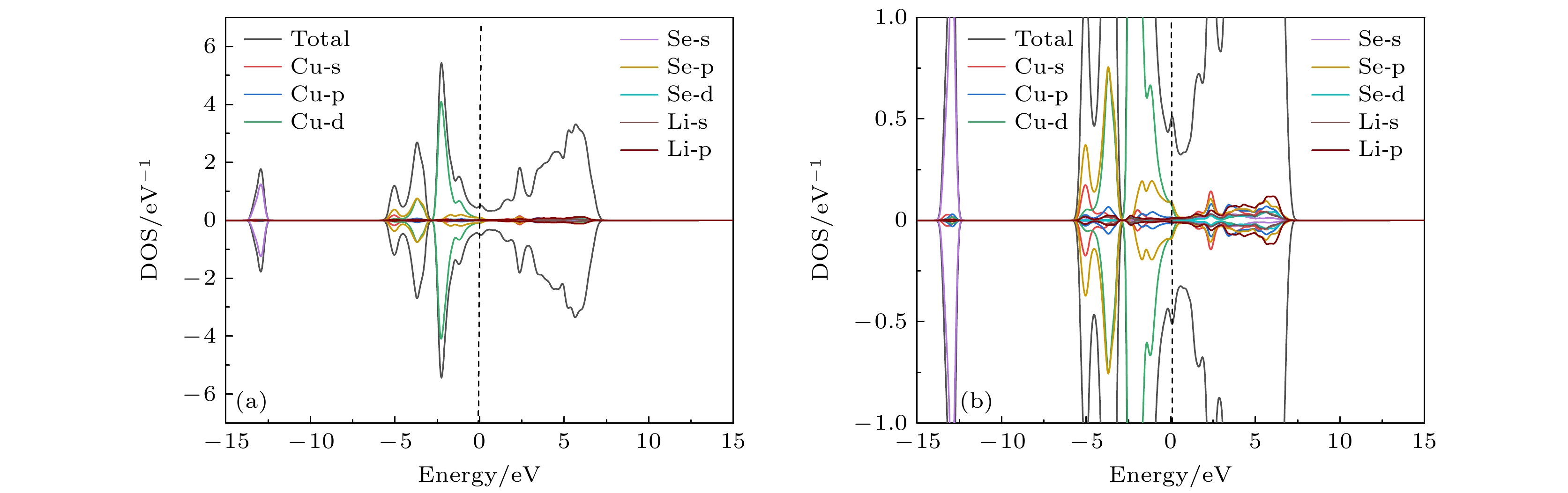
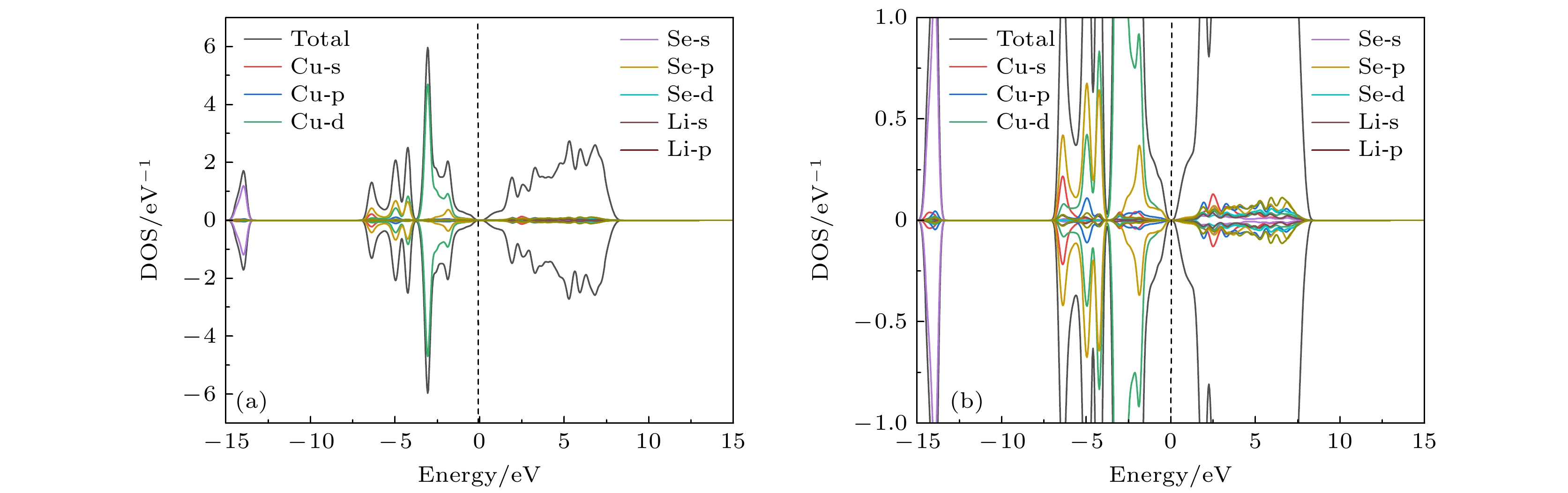
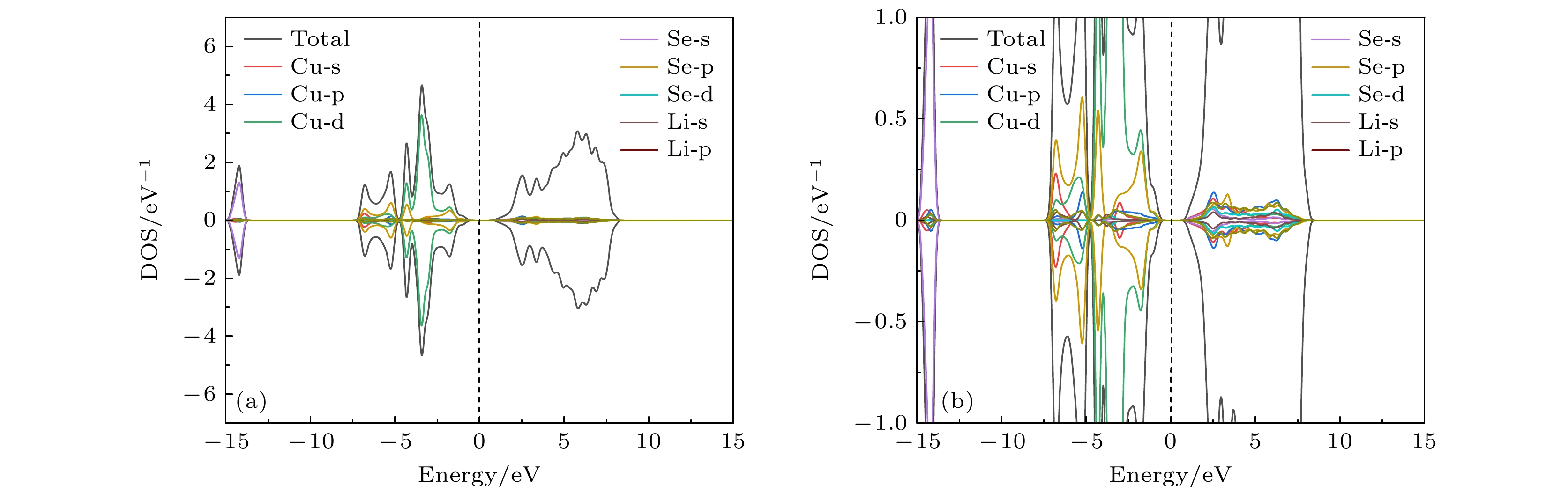
Original bulk phases of two-dimensional atomic crystal materials are layered. However, a few relevant researches show that some of two-dimensional material crystals have non-layered bulk phases. In this work we investigate monolayer CuSe which is non-layered, belonging in a new kind of honeycomb graphene analogue. Monolayer CuSe is not suitable for application in electronic devices because of its metallic nature. In order to find new two-dimensional atomic crystal materials with excellent performance suitable for application in electronic devices, we change CuSe from metal to semiconductor through external atom modification. The first principles study of density functional theory is conducted to ascertain the energy band structure of monolayer CuSe after second periodic atoms have been added to the top, center and bridge sites. The characteristics of monolayer CuSe with addition of Li or B atoms are studied, including energy band structure, the density of states, differential charge density, and crystal orbital Hamiltonian population. The results show that after adding Li atoms to CuSe, the CuSe transforms from metallic to semiconductive property at all three positions, and Li atom is more easily to be modified in the hexagonal center of CuSe, with band gap being about 1.77 eV, the Fermi level biased towards the top of the valence band. The CuSe with addition of Li atoms exhibits a p-type semiconductor property, so it is a direct bandgap semiconductor. Adding B atom to the top of Cu atom can also make CuSe semiconductive, with a band gap of about 1.2 eV, the conduction band minimum at the K point, and the valence band maximum at the Γ point. The CuSe with addition of B atoms belongs in an indirect band gap semiconductor, and the Fermi energy level is biased towards the conduction band minimum, exhibiting the characteristics of an n-type semiconductor. According to the results of differential charge density and crystal orbital Hamiltonian population, the B atom is bound to the top of the monolayer CuSe with the B-Se polar covalent bond. The first principle study reveals the realization of metal-to-semiconductor transition from monolayer CuSe to CuXSe (X = Li, B), and the calculation results also show that CuSe with addition of Li atoms or B atoms is likely to be used in future electronic devices.
-
Keywords:
- monolayer CuSe /
- first-principles calculations /
- electronic structure
[1] Novoselov K S, Geim A K, Morozov S V, Jiang D, Zhang Y, Dubonos S V, Grigorieva I V, Firsov A A 2004 Science 306 666
 Google Scholar
Google Scholar
[2] Zhao J J, Liu H S, Yu Z M, Quhe R, Zhou S, Wang Y Y, Liu C C, Zhong H X, Han N N, Lu J, Yao Y G, Wu K H 2016 Prog. Mater. Sci. 83 24
 Google Scholar
Google Scholar
[3] Lang J L, Ding B, Zhang S, Su H X, Ge B H, Qi L H, Gao H J, Li X Y, Li Q Y, Wu H 2017 Adv. Mater. 29 1701777
 Google Scholar
Google Scholar
[4] Feng B J, Ding Z J, Meng S, Yao Y G, He X Y, Cheng P, Chen L, Wu K H 2012 Nano Lett. 12 3507
 Google Scholar
Google Scholar
[5] Liao Y L, Chen Z F, Connell J W, Fay C C, Park C, Kim J W, Lin Y 2014 Adv. Funct. Mater. 24 4497
 Google Scholar
Google Scholar
[6] Zeng H B, Zhi C Y, Zhang Z H, Wei X L, Wang X B, Guo W L, Bando Y, Golberg D 2010 Nano Lett. 10 5049
 Google Scholar
Google Scholar
[7] Kumar R, Sahoo S, Joanni E, Singh R K, Yadav R M, Verma R K, Singh D P, Tan W K, Pino A P, Moshkalev S A, Matsuda A 2019 Nano Res. 12 2655
 Google Scholar
Google Scholar
[8] Splendiani A, Sun L, Zhang Y B, Li T S, Kim J, Chim C Y, Galli G, Wang F 2010 Nano Lett. 10 1271
 Google Scholar
Google Scholar
[9] Mak K F, Lee C, Hone J, Shan J, Heinz T F 2010 Phys. Rev. Lett. 105 136805
 Google Scholar
Google Scholar
[10] Chhowalla M, Liu Z F, Zhang H 2015 Chem. Soc. Rev. 44 2584
 Google Scholar
Google Scholar
[11] Naguib M, Kurtoglu M, Presser V, Lu J, Niu J J, Heon M, Hultman L, Gogotsi Y, Barsoum M W 2011 Adv. Mater. 23 4248
 Google Scholar
Google Scholar
[12] Naguib M, Mashtalir O, Carle J, Presser V, Lu J, Hultman L, Gogotsi Y, Barsoum M W 2012 Acs Nano 6 1322
 Google Scholar
Google Scholar
[13] Zhan X X, Si C, Zhou J, Sun Z M 2020 Nano. Horiz. 5 235
 Google Scholar
Google Scholar
[14] Aydın Z Y, Abacı S 2017 Solid State Sci. 74 74
 Google Scholar
Google Scholar
[15] Buffiere M, Dhawale D S, EI-Mellouhi F 2019 Energy Technol. 7 1900819
 Google Scholar
Google Scholar
[16] Yang Z Q, Wang S C, Li H L, Yang J P, Zhao J X, Qu W Q, Shih K 2020 Ind. End. Chem. Res. 59 13603
 Google Scholar
Google Scholar
[17] Masrat S, Poolla R, Dipak P, Zaman M B 2021 Surf. Interfaces 23 100973
 Google Scholar
Google Scholar
[18] Cheng Y S, Zhang J, Xiong X S, Chen C, Zeng J H, Kong Z, Wang H B, Xi J H, Yuan Y J, Ji Z G 2021 J. Alloy. Compd. 870 159540
 Google Scholar
Google Scholar
[19] Weng J H, Gao S P 2019 Rsc. Adv. 9 32984
 Google Scholar
Google Scholar
[20] Weng J H, Gao S P 2021 J. Phys. Chem. Solids 148 109738
 Google Scholar
Google Scholar
[21] Yang G, Xu W X, Gao S P 2021 Comput. Mater. Sci. 198 110696
 Google Scholar
Google Scholar
[22] Ruffieux P, Wang S Y, Yang B, Sanchez-Sanchez C, Liu J, Dienel T, Talirz L, Shinde P, Pignedoli C A, Passerone D, Dumslaff T, Feng X L, Mullen K, Fasel R 2016 Nature 531 489
 Google Scholar
Google Scholar
[23] Nakanishi T, Ando T 2015 Phys. Rev. B 91 155420
 Google Scholar
Google Scholar
[24] Chamlagain B, Withanage S S, Johnston A C, Khondaker S I 2020 Sci. Rep. 10 12970
 Google Scholar
Google Scholar
[25] Cao T, Li Z L, Louie S G 2015 Phys. Rev. Lett. 114 236602
 Google Scholar
Google Scholar
[26] Kang M G, Kim B, Ryu S H, Jung S W, Kim J, Moreschini L, Jozwiak C, Rotenberg E, Bostwick A, Kim K S 2017 Nano Lett. 17 1610
 Google Scholar
Google Scholar
[27] Dai Z H, Liu L Q, Zhang Z 2019 Adv. Mater. 31 1805417
 Google Scholar
Google Scholar
[28] Cui X, Lee G H, Kim Y D, Arefe G, Huang P Y, Lee C H, Chenet D A, Zhang X, Wang L, Ye F, Pizzocchero F, Jessen B S, Watanabe K, Taniguchi T, Muller D A, Low T, Kim P, Hone J 2015 Nat. Nanotech. 10 534
 Google Scholar
Google Scholar
[29] Ju L, Shi Z W, Nair N, Lü Y C, Jin C H, Jr J V, Ojeda-Aristizabal C, Bechtel H A, Martin M C, Zettl A, Analytis J and Wang F 2015 Nature 520 650
 Google Scholar
Google Scholar
[30] Lebegue S, Klintenberg M, Eriksson O, Katsnelson M I 2009 Phys. Rev. B 79 245117
 Google Scholar
Google Scholar
[31] Yang J H, Song S R, Du S X, Gao H J, Yakobson B I 2017 J. Phys. Chem. Lett. 8 4594
 Google Scholar
Google Scholar
[32] Wang Q C, Lei Y P, Wang Y C, Liu Y, Song C Y, Zeng J, Song Y H, Duan X D, Wang D S, Li Y D 2020 Energy Environ. Sci. 13 1593
 Google Scholar
Google Scholar
[33] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[34] Perdew J P, Burke K, Ernzerhof M 1996 Phy. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[35] Wu X J, Huang X, Liu J Q, Li H, Yang J, Li B, Huang W, Zhang H 2014 Angew. Chem. 126 5183
 Google Scholar
Google Scholar
-
图 3 CuXSe (X = Li, Be, B, C, N, O, F)的能带结构 (a)—(c), (d)—(f), (g)—(i), (j)—(l), (m)—(o), (p)—(r), (s)—(u) 依次表示Li, Be, B, C, N, O, F原子分别在CuSe的顶部位置、中心位置和桥位
Figure 3. Energy band structure of CuXSe (X = Li, Be, B, C, N, O, F): (a)–(c), (d)–(f), (g)–(i), (j)–(l), (m)–(o), (p)–(r), (s)–(u) Indicate the top position, center position, and bridge position of Li, Be, B, C, N, O and F atoms in CuSe in sequence.
表 1 CuXSe (X = Li, Be, B, C, N, O, F)体系不同位置的形成能
Table 1. Formation energy at different positions of CuXSe (X = Li, Be, B, C, N, O, F)system.
掺杂体系 不同位置的形成能$ {E}_{{\rm{f}}} $/eV Cu原子的顶位 Se原子的顶位 中心位置 桥位 最稳定的位置 CuLiSe –2.016 –1.747 –2.622 –2.257 中心位置 CuBeSe –2.456 –1.979 –2.091 –2.520 桥位 CuBSe –3.509 –3.723 –2.484 –2.121 Se原子的顶位 CuCSe –4.291 –4.212 –2.619 –2.452 Cu原子的顶位 CuNSe –0.860 –1.647 –1.044 –1.532 Se原子的顶位 CuOSe –3.271 –2.977 –1.704 –2.177 Cu原子的顶位 CuFSe –2.537 –2.737 –1.398 –2.087 Se原子的顶位 -
[1] Novoselov K S, Geim A K, Morozov S V, Jiang D, Zhang Y, Dubonos S V, Grigorieva I V, Firsov A A 2004 Science 306 666
 Google Scholar
Google Scholar
[2] Zhao J J, Liu H S, Yu Z M, Quhe R, Zhou S, Wang Y Y, Liu C C, Zhong H X, Han N N, Lu J, Yao Y G, Wu K H 2016 Prog. Mater. Sci. 83 24
 Google Scholar
Google Scholar
[3] Lang J L, Ding B, Zhang S, Su H X, Ge B H, Qi L H, Gao H J, Li X Y, Li Q Y, Wu H 2017 Adv. Mater. 29 1701777
 Google Scholar
Google Scholar
[4] Feng B J, Ding Z J, Meng S, Yao Y G, He X Y, Cheng P, Chen L, Wu K H 2012 Nano Lett. 12 3507
 Google Scholar
Google Scholar
[5] Liao Y L, Chen Z F, Connell J W, Fay C C, Park C, Kim J W, Lin Y 2014 Adv. Funct. Mater. 24 4497
 Google Scholar
Google Scholar
[6] Zeng H B, Zhi C Y, Zhang Z H, Wei X L, Wang X B, Guo W L, Bando Y, Golberg D 2010 Nano Lett. 10 5049
 Google Scholar
Google Scholar
[7] Kumar R, Sahoo S, Joanni E, Singh R K, Yadav R M, Verma R K, Singh D P, Tan W K, Pino A P, Moshkalev S A, Matsuda A 2019 Nano Res. 12 2655
 Google Scholar
Google Scholar
[8] Splendiani A, Sun L, Zhang Y B, Li T S, Kim J, Chim C Y, Galli G, Wang F 2010 Nano Lett. 10 1271
 Google Scholar
Google Scholar
[9] Mak K F, Lee C, Hone J, Shan J, Heinz T F 2010 Phys. Rev. Lett. 105 136805
 Google Scholar
Google Scholar
[10] Chhowalla M, Liu Z F, Zhang H 2015 Chem. Soc. Rev. 44 2584
 Google Scholar
Google Scholar
[11] Naguib M, Kurtoglu M, Presser V, Lu J, Niu J J, Heon M, Hultman L, Gogotsi Y, Barsoum M W 2011 Adv. Mater. 23 4248
 Google Scholar
Google Scholar
[12] Naguib M, Mashtalir O, Carle J, Presser V, Lu J, Hultman L, Gogotsi Y, Barsoum M W 2012 Acs Nano 6 1322
 Google Scholar
Google Scholar
[13] Zhan X X, Si C, Zhou J, Sun Z M 2020 Nano. Horiz. 5 235
 Google Scholar
Google Scholar
[14] Aydın Z Y, Abacı S 2017 Solid State Sci. 74 74
 Google Scholar
Google Scholar
[15] Buffiere M, Dhawale D S, EI-Mellouhi F 2019 Energy Technol. 7 1900819
 Google Scholar
Google Scholar
[16] Yang Z Q, Wang S C, Li H L, Yang J P, Zhao J X, Qu W Q, Shih K 2020 Ind. End. Chem. Res. 59 13603
 Google Scholar
Google Scholar
[17] Masrat S, Poolla R, Dipak P, Zaman M B 2021 Surf. Interfaces 23 100973
 Google Scholar
Google Scholar
[18] Cheng Y S, Zhang J, Xiong X S, Chen C, Zeng J H, Kong Z, Wang H B, Xi J H, Yuan Y J, Ji Z G 2021 J. Alloy. Compd. 870 159540
 Google Scholar
Google Scholar
[19] Weng J H, Gao S P 2019 Rsc. Adv. 9 32984
 Google Scholar
Google Scholar
[20] Weng J H, Gao S P 2021 J. Phys. Chem. Solids 148 109738
 Google Scholar
Google Scholar
[21] Yang G, Xu W X, Gao S P 2021 Comput. Mater. Sci. 198 110696
 Google Scholar
Google Scholar
[22] Ruffieux P, Wang S Y, Yang B, Sanchez-Sanchez C, Liu J, Dienel T, Talirz L, Shinde P, Pignedoli C A, Passerone D, Dumslaff T, Feng X L, Mullen K, Fasel R 2016 Nature 531 489
 Google Scholar
Google Scholar
[23] Nakanishi T, Ando T 2015 Phys. Rev. B 91 155420
 Google Scholar
Google Scholar
[24] Chamlagain B, Withanage S S, Johnston A C, Khondaker S I 2020 Sci. Rep. 10 12970
 Google Scholar
Google Scholar
[25] Cao T, Li Z L, Louie S G 2015 Phys. Rev. Lett. 114 236602
 Google Scholar
Google Scholar
[26] Kang M G, Kim B, Ryu S H, Jung S W, Kim J, Moreschini L, Jozwiak C, Rotenberg E, Bostwick A, Kim K S 2017 Nano Lett. 17 1610
 Google Scholar
Google Scholar
[27] Dai Z H, Liu L Q, Zhang Z 2019 Adv. Mater. 31 1805417
 Google Scholar
Google Scholar
[28] Cui X, Lee G H, Kim Y D, Arefe G, Huang P Y, Lee C H, Chenet D A, Zhang X, Wang L, Ye F, Pizzocchero F, Jessen B S, Watanabe K, Taniguchi T, Muller D A, Low T, Kim P, Hone J 2015 Nat. Nanotech. 10 534
 Google Scholar
Google Scholar
[29] Ju L, Shi Z W, Nair N, Lü Y C, Jin C H, Jr J V, Ojeda-Aristizabal C, Bechtel H A, Martin M C, Zettl A, Analytis J and Wang F 2015 Nature 520 650
 Google Scholar
Google Scholar
[30] Lebegue S, Klintenberg M, Eriksson O, Katsnelson M I 2009 Phys. Rev. B 79 245117
 Google Scholar
Google Scholar
[31] Yang J H, Song S R, Du S X, Gao H J, Yakobson B I 2017 J. Phys. Chem. Lett. 8 4594
 Google Scholar
Google Scholar
[32] Wang Q C, Lei Y P, Wang Y C, Liu Y, Song C Y, Zeng J, Song Y H, Duan X D, Wang D S, Li Y D 2020 Energy Environ. Sci. 13 1593
 Google Scholar
Google Scholar
[33] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[34] Perdew J P, Burke K, Ernzerhof M 1996 Phy. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[35] Wu X J, Huang X, Liu J Q, Li H, Yang J, Li B, Huang W, Zhang H 2014 Angew. Chem. 126 5183
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 7767
- PDF Downloads: 242
- Cited By: 0















 DownLoad:
DownLoad: