-
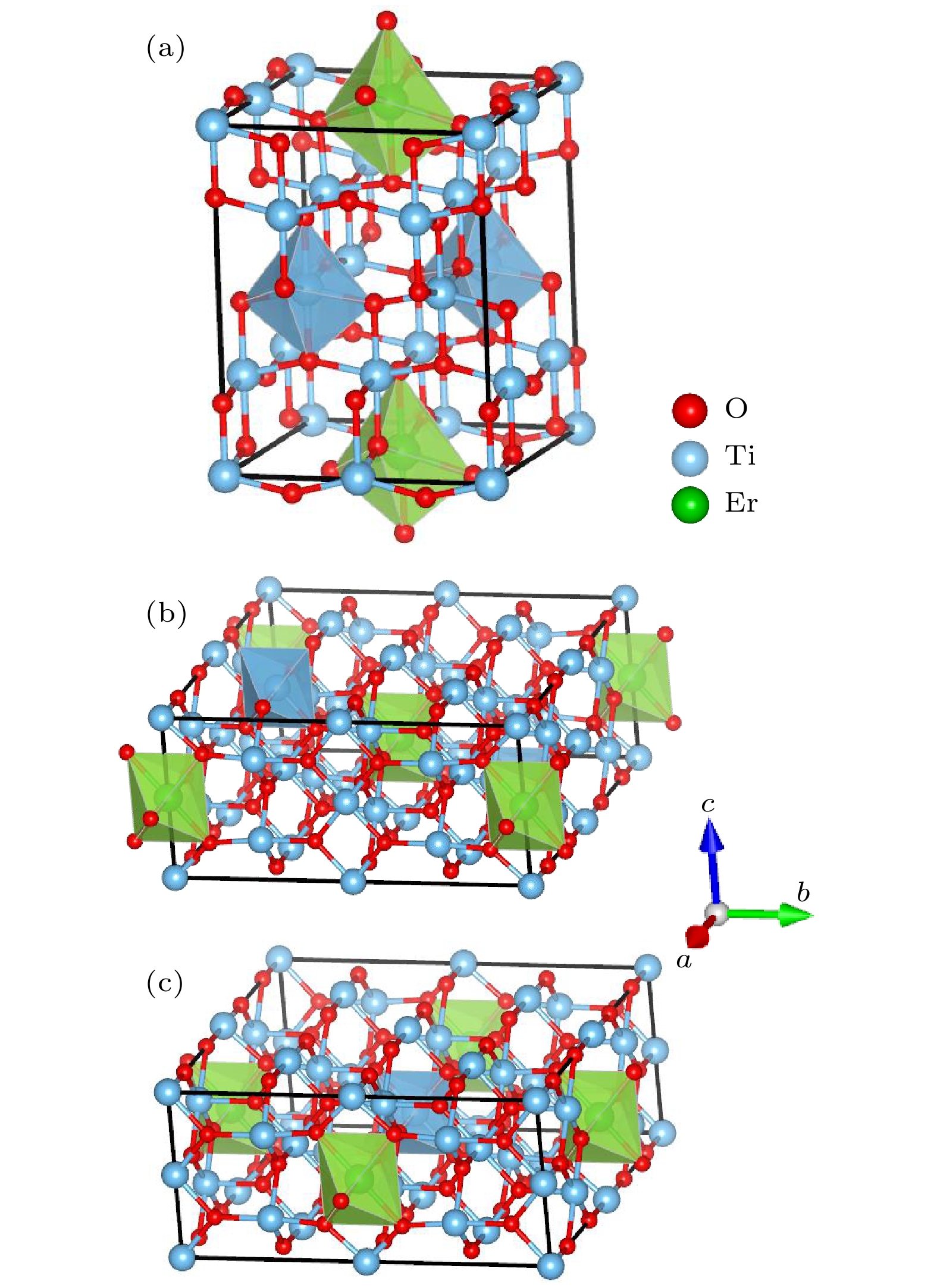
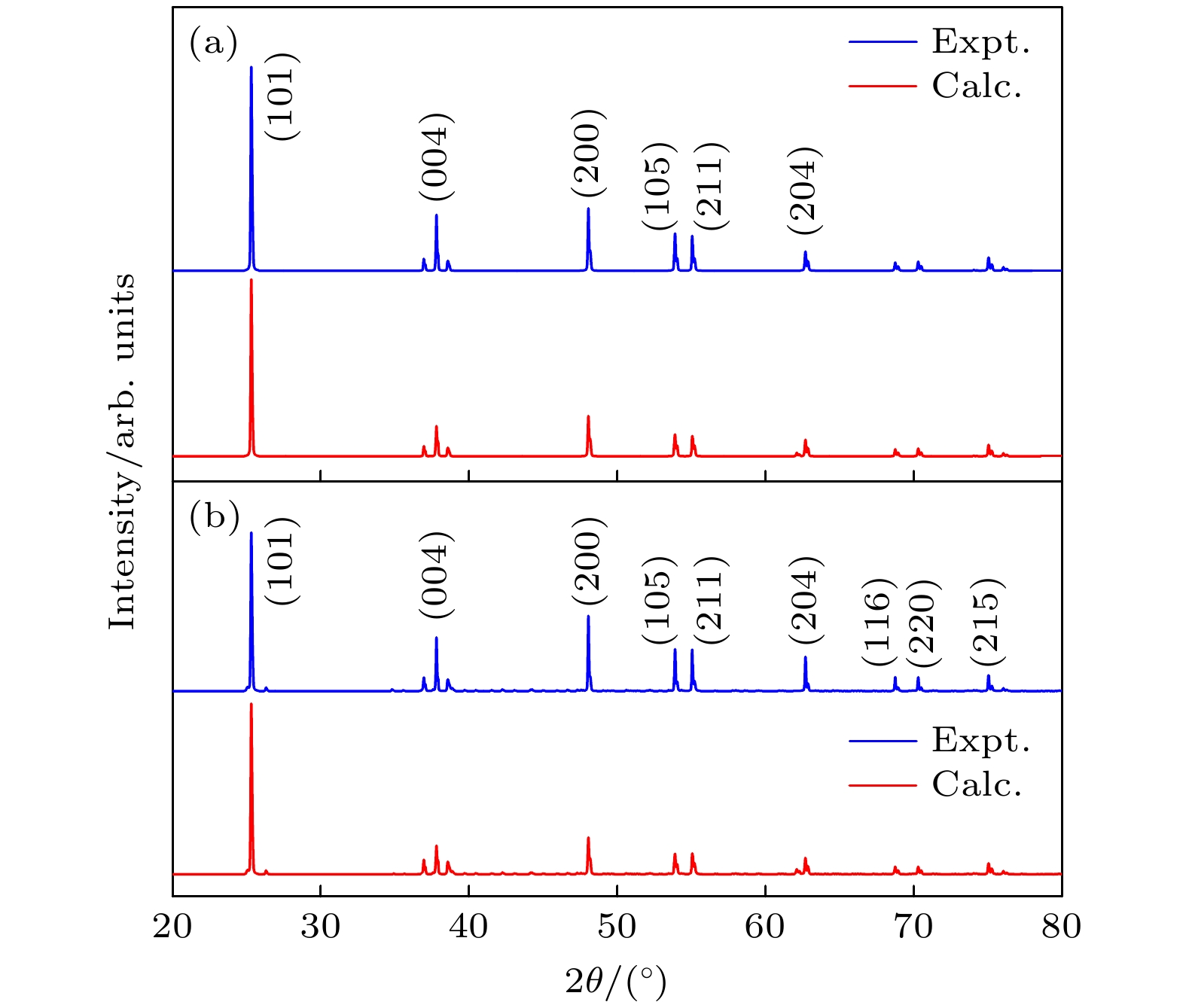
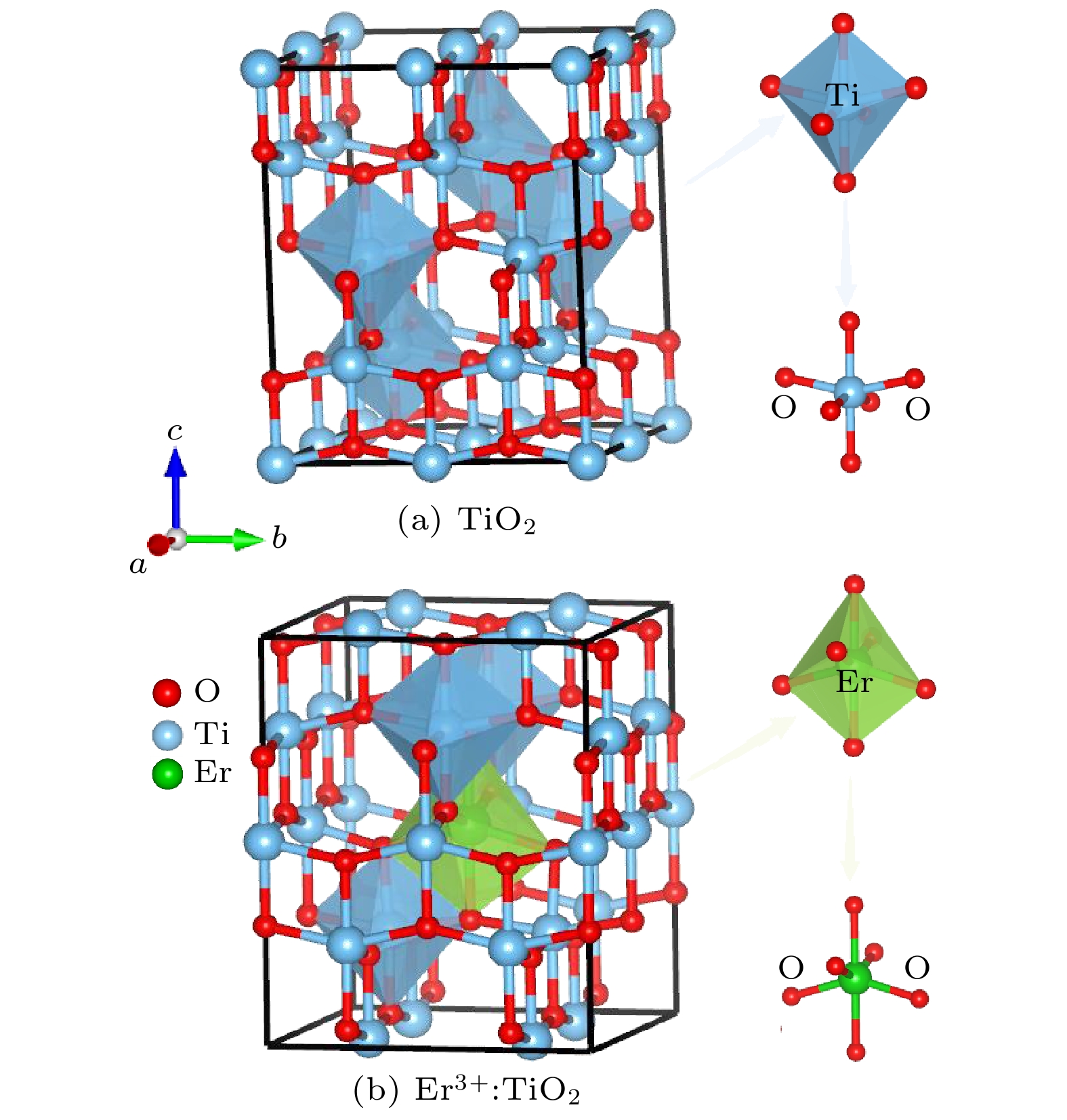
Trivalent rare earth erbium ion (Er3+) doped titanium oxide (TiO2) can possess a very wide range of applications due to its excellent optoelectronic properties, thus standing out among many rare-earth-doped luminescent crystals. However, the issues regarding local structure and electronic properties have not been finalized. To address these problems, the CALYPSO (Crystal structure AnaLYsis by Particle Swarm Optimization) method combined with the first-principles calculations is employed, and many converged structures of Er3+-doped TiO2 are successfully obtained. Further structural optimization is performed by using the VASP (Vienna ab initio simulation package) software package, and we report for the first time that the lowest energy structure of Er3+-doped TiO2 has the
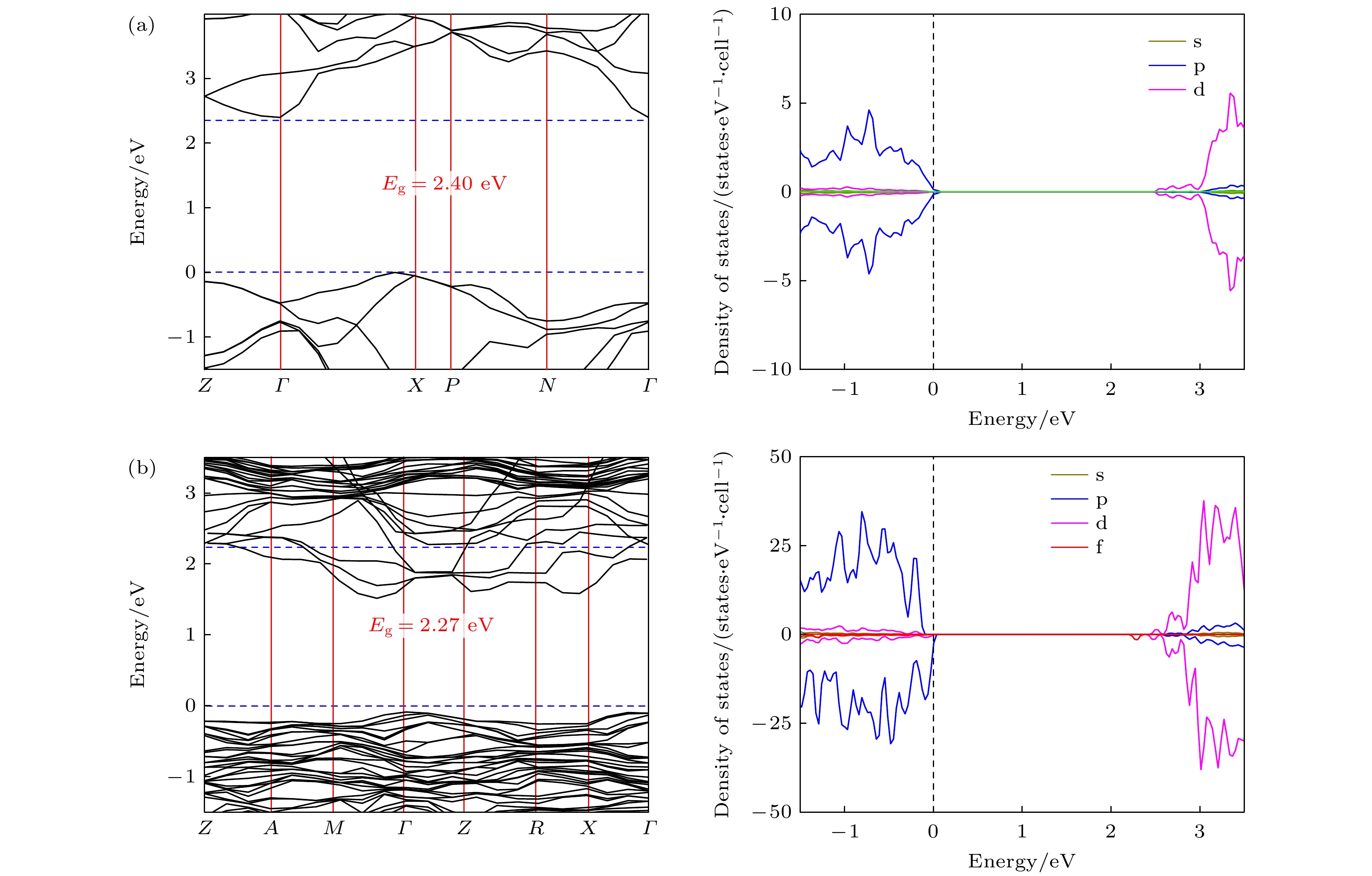
$ P\overline 4 $ m2 symmetry. It can be observed that the doped Er3+ ions enter into the host crystal and occupy the positions of Ti4+ ions, resulting in structural distortion, which eventually leads the local Er3+ coordination site symmetry to reduce from D2d into C2v. We speculate that there are two reasons: 1) the difference in charge between Er3+ ions and Ti4+ ions leads to charge compensation; 2) the difference between their electron radii is obvious: the radius is 0.0881 for Er3+ ion and 0.0881 for Ti4+ ion. In addition, during the structural search, we also find many metastable structures that may exist at a special temperature or pressure, which play an important role in the studying of structural evolution. When the electronic band structure of the Er3+-doped TiO2 system is calculated, we adopt the method of local density approximation (LDA) combined with the on-site Coulomb repulsion parameter U to accurately describe the strongly correlated system. For the specific value of U, we adopt 3.5 eV and 7.6 eV to describe the strong correlation of 3d electrons of Ti4+ ions and 4f electrons of Er3+ ions, respectively. According to the calculation of electronic properties, the band gap value of Er3+ doped TiO2 is about 2.27 eV, which is lower than that of the host crystal (Eg = 2.40 eV). The results show that the reduction in the band gap is mainly caused by the f state of Er3+ ions. The doping of Er ion does reduce the band gap value, but it does not change the conductivity of the system, which have great application prospect in diode-pumped laser. These findings not only provide the data for further exploring the properties and applications of Er3+:TiO2 crystals, but also present an approach to studying other rare-earth-doped crystalline materials.-
Keywords:
- CALYPSO /
- Er3+-doped TiO2 /
- local structure /
- first-principles calculations
[1] 钟淑琳, 仇家豪, 罗文崴, 吴木生 2021 70 158203
 Google Scholar
Google Scholar
Zhong S L, Qiu J H, Luo W W, Wu M S 2021 Acta Phys. Sin. 70 158203
 Google Scholar
Google Scholar
[2] 孟勇军, 李洪, 唐建伟, 陈学文 2022 71 027801
 Google Scholar
Google Scholar
Meng Y J, Li H, Tang J W, Chen X W 2022 Acta Phys. Sin. 71 027801
 Google Scholar
Google Scholar
[3] Wu B, Zhao L, Wang Y, Dong H, Yu H 2019 RSC Adv. 9 42228
 Google Scholar
Google Scholar
[4] Menezes L D S, Araújo C B D 2015 J. Braz. Chem. Soc. 26 2405
[5] Stengl V, Bakardjieva S, Murafa N 2009 Mater. Chem. Phys. 114 217
 Google Scholar
Google Scholar
[6] Hassan M S, Amna T, Yang O B, Kim H C, Khil M S 2012 Ceram. Int. 38 5925
 Google Scholar
Google Scholar
[7] Borlaf M, Colomer M T, Moreno R, Ortiz A L 2014 J. Eur. Ceram. Soc. 34 4457
 Google Scholar
Google Scholar
[8] Bao R, Li R, Chen C, Wu H, Xia J, Long C, Li H 2019 J. Phys. Chem. Solid. 126 78
 Google Scholar
Google Scholar
[9] Li J G, Wang X H, Kamiyama H, Ishigaki T, Sekiguchi T 2006 Thin Solid Films 506 292
[10] Agrios A G, Pochat P 2005 J. Appl. Electrochem. 35 655
 Google Scholar
Google Scholar
[11] Camps I, Borlaf M, Toudert J, Andres A D, Colomer M T, Moreno R, Serna R 2018 J. Alloys Compd. 735 2267
 Google Scholar
Google Scholar
[12] Talane T E 2018 M. S. Thesis (Gauteng Province: University of South Africa)
[13] Pablo L I, Laeticia P, Jonathan M, Davide J, Nadia G B, Diego P, Sonia F, Chiara N, Fabrizio G, Daniel M 2017 J. Non-Cryst. Solids 460 161
 Google Scholar
Google Scholar
[14] Mignotte C 2004 Appl. Surf. Sci. 226 355
 Google Scholar
Google Scholar
[15] Talane T E, Mbule P S, Noto L L, Shingange K, Mhlongo G H, Mothudi B M, Dhlamini M S 2018 Mater. Res. Bull. 108 234
 Google Scholar
Google Scholar
[16] Wild J D, Meijerink A, Rath J K, van Sark W G J H M, Schropp R E I 2011 Energy Environ. Sci. 4 4835
 Google Scholar
Google Scholar
[17] Pablo L I, Diego P, Nadia G B, Davide J, Giovanni B, Laeticia P, Daniel M 2018 Nanomaterials 8 20
 Google Scholar
Google Scholar
[18] van den Hoven G N, Koper R J I M, Polman A, Dam C V, Uffelen J W M V, Smit M K 1996 Appl. Phys. Lett. 68 1886
 Google Scholar
Google Scholar
[19] Jia C W, Zhao J G, Duan H G, Xie E Q 2007 Mater. Lett. 61 4389
 Google Scholar
Google Scholar
[20] Fu C Y, Liao J S, Luo W Q, Li R F, Chen X Y 2008 Opt. Lett. 33 953
 Google Scholar
Google Scholar
[21] Luo W Q, Fu C Y, Li R F, Liu Y S, Zhu H M, Chen X Y 2011 Small 7 3046
 Google Scholar
Google Scholar
[22] Ren Z, Wu J, Wang N, Li X 2018 J. Mater. Chem. A 6 15348
 Google Scholar
Google Scholar
[23] Mazierski P, Mikolajczyk A, Grzybd T, Caicedo P N A, Wei Z, Kowalska E, Henry P P, Adriana Z M, Nadolna J 2020 Appl. Surf. Sci. 527 146815
 Google Scholar
Google Scholar
[24] Wang Y C, Lv J, Zhu L, Ma Y M 2012 Comput. Phys. Commun. 183 2063
 Google Scholar
Google Scholar
[25] Wang Y C, Lv J, Zhu L, Lu S H, Yin K T, Li Q, Wang H, Zhang L J, Ma Y M 2015 J. Phys. Condens. Matter. 27 203203
 Google Scholar
Google Scholar
[26] Wang H, Wang Y C, Lv J, Li Q, Zhang L J, Ma Y M 2016 Comput. Mater. Sci. 112 406
 Google Scholar
Google Scholar
[27] Wang Y C, Lv J, Zhu L, Ma Y M 2010 Phys. Rev. B 82 094116
 Google Scholar
Google Scholar
[28] Gao B, Gao P, Lu S, Lv J, Wang Y, Ma Y 2019 Sci. Bull. 64 301
 Google Scholar
Google Scholar
[29] Wang Y, Miao M, Lv J, Zhu L, Yin K, Liu H, Ma Y 2012 J. Chem. Phys. 137 224108
 Google Scholar
Google Scholar
[30] Hafner J 2008 J. Comput. Chem. 29 2044
 Google Scholar
Google Scholar
[31] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[32] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[33] Sanna S, Schmidt W G, Frauenheim T, Gerstmann U 2009 Phys. Rev. B 80 104120
 Google Scholar
Google Scholar
[34] Xiao Y, Ju M, Yuan H K, Yeung Y Y 2021 J. Phys. Chem. C 125 18015
 Google Scholar
Google Scholar
[35] Togo A, Tanaka I 2015 Scr. Mater. 108 1
 Google Scholar
Google Scholar
[36] Phenicie C M, Stevenson P, Welinski S, Rose B C, Asfaw A T, Cava R J, Lyon S A, de Leon N P, Thompson J D 2019 Nano Lett. 19 8928
 Google Scholar
Google Scholar
[37] Mills A, Hunte S L 1997 J. Photochem. Photobiol. A 108 1
 Google Scholar
Google Scholar
[38] Yang J, Hu Y, Jin C, Zhuge L, Wu X 2017 Thin Solid Films 637 9
 Google Scholar
Google Scholar
[39] Pan L, Xiao Y, Kuang X Y, Ju M 2021 Mater. Chem. Phys. 257 123824
 Google Scholar
Google Scholar
[40] Savin A, Nesper R, Wengert S, Fässler T F 1997 Angew. Chem. Int. Ed. Engl. 36 1808
 Google Scholar
Google Scholar
[41] Lu T, Chen F 2011 Acta Phys. Chim. Sin. 27 2786
 Google Scholar
Google Scholar
[42] Fuentealba P, Chamorro E, Santos J C 2007 Theoretical Aspects of Chemical Reactivity 19 57
-
表 1 Er3+:TiO2能量最低结构中所有原子的坐标
Table 1. Coordinates of all atoms for the low-energy structure of Er3+:TiO2.
Atom x y z Wyckoff site
symmetryO(1) 0.247 0.249 0.204 4j O(2) 0.753 0.751 0.204 4k O(3) 0.249 0.753 0.796 2g O(4) 0.751 0.247 0.796 8l O(6) 0.753 0.249 0.204 2g O(7) 0.250 0.247 0.796 4k O(8) 0.751 0.753 0.796 4j O(11) 0.250 0.000 0.545 2f O(19) 0.500 0.753 0.036 2e Ti(1) 0.000 0.250 0.251 4j Ti(2) 0.000 0.750 0.251 4k Ti(3) 0.250 0.000 0.749 2g Ti(4) 0.750 0.000 0.749 4h Ti(10) 0.500 0.000 0.495 1d Er(1) 0.500 0.500 0.500 1c 表 2 Er3+:TiO2的基态结构以及亚稳态结构的晶格参数a, b, c, 原胞体积V, 相对能量∆E
Table 2. Structural parameters a, b and c, unit-cell volume, relative energies for the optimized TiO2 and metastable Er3+:TiO2
Space group a/Å b/Å c/Å V/Å3 ∆E/eV TiO2 I41/amd 7.568 7.568 9.515 545.003 — Er3+:TiO2 $ P\overline 4 $m2 7.682 7.682 9.798 578.193 0 Isomer (a) $ P\overline 4 $m2 7.681 7.681 9.799 578.213 0.051 Isomer (b) Cmmm 13.258 13.258 6.013 1051.764 0.853 Isomer (c) Cmmm 13.261 13.261 6.009 1051.988 0.975 -
[1] 钟淑琳, 仇家豪, 罗文崴, 吴木生 2021 70 158203
 Google Scholar
Google Scholar
Zhong S L, Qiu J H, Luo W W, Wu M S 2021 Acta Phys. Sin. 70 158203
 Google Scholar
Google Scholar
[2] 孟勇军, 李洪, 唐建伟, 陈学文 2022 71 027801
 Google Scholar
Google Scholar
Meng Y J, Li H, Tang J W, Chen X W 2022 Acta Phys. Sin. 71 027801
 Google Scholar
Google Scholar
[3] Wu B, Zhao L, Wang Y, Dong H, Yu H 2019 RSC Adv. 9 42228
 Google Scholar
Google Scholar
[4] Menezes L D S, Araújo C B D 2015 J. Braz. Chem. Soc. 26 2405
[5] Stengl V, Bakardjieva S, Murafa N 2009 Mater. Chem. Phys. 114 217
 Google Scholar
Google Scholar
[6] Hassan M S, Amna T, Yang O B, Kim H C, Khil M S 2012 Ceram. Int. 38 5925
 Google Scholar
Google Scholar
[7] Borlaf M, Colomer M T, Moreno R, Ortiz A L 2014 J. Eur. Ceram. Soc. 34 4457
 Google Scholar
Google Scholar
[8] Bao R, Li R, Chen C, Wu H, Xia J, Long C, Li H 2019 J. Phys. Chem. Solid. 126 78
 Google Scholar
Google Scholar
[9] Li J G, Wang X H, Kamiyama H, Ishigaki T, Sekiguchi T 2006 Thin Solid Films 506 292
[10] Agrios A G, Pochat P 2005 J. Appl. Electrochem. 35 655
 Google Scholar
Google Scholar
[11] Camps I, Borlaf M, Toudert J, Andres A D, Colomer M T, Moreno R, Serna R 2018 J. Alloys Compd. 735 2267
 Google Scholar
Google Scholar
[12] Talane T E 2018 M. S. Thesis (Gauteng Province: University of South Africa)
[13] Pablo L I, Laeticia P, Jonathan M, Davide J, Nadia G B, Diego P, Sonia F, Chiara N, Fabrizio G, Daniel M 2017 J. Non-Cryst. Solids 460 161
 Google Scholar
Google Scholar
[14] Mignotte C 2004 Appl. Surf. Sci. 226 355
 Google Scholar
Google Scholar
[15] Talane T E, Mbule P S, Noto L L, Shingange K, Mhlongo G H, Mothudi B M, Dhlamini M S 2018 Mater. Res. Bull. 108 234
 Google Scholar
Google Scholar
[16] Wild J D, Meijerink A, Rath J K, van Sark W G J H M, Schropp R E I 2011 Energy Environ. Sci. 4 4835
 Google Scholar
Google Scholar
[17] Pablo L I, Diego P, Nadia G B, Davide J, Giovanni B, Laeticia P, Daniel M 2018 Nanomaterials 8 20
 Google Scholar
Google Scholar
[18] van den Hoven G N, Koper R J I M, Polman A, Dam C V, Uffelen J W M V, Smit M K 1996 Appl. Phys. Lett. 68 1886
 Google Scholar
Google Scholar
[19] Jia C W, Zhao J G, Duan H G, Xie E Q 2007 Mater. Lett. 61 4389
 Google Scholar
Google Scholar
[20] Fu C Y, Liao J S, Luo W Q, Li R F, Chen X Y 2008 Opt. Lett. 33 953
 Google Scholar
Google Scholar
[21] Luo W Q, Fu C Y, Li R F, Liu Y S, Zhu H M, Chen X Y 2011 Small 7 3046
 Google Scholar
Google Scholar
[22] Ren Z, Wu J, Wang N, Li X 2018 J. Mater. Chem. A 6 15348
 Google Scholar
Google Scholar
[23] Mazierski P, Mikolajczyk A, Grzybd T, Caicedo P N A, Wei Z, Kowalska E, Henry P P, Adriana Z M, Nadolna J 2020 Appl. Surf. Sci. 527 146815
 Google Scholar
Google Scholar
[24] Wang Y C, Lv J, Zhu L, Ma Y M 2012 Comput. Phys. Commun. 183 2063
 Google Scholar
Google Scholar
[25] Wang Y C, Lv J, Zhu L, Lu S H, Yin K T, Li Q, Wang H, Zhang L J, Ma Y M 2015 J. Phys. Condens. Matter. 27 203203
 Google Scholar
Google Scholar
[26] Wang H, Wang Y C, Lv J, Li Q, Zhang L J, Ma Y M 2016 Comput. Mater. Sci. 112 406
 Google Scholar
Google Scholar
[27] Wang Y C, Lv J, Zhu L, Ma Y M 2010 Phys. Rev. B 82 094116
 Google Scholar
Google Scholar
[28] Gao B, Gao P, Lu S, Lv J, Wang Y, Ma Y 2019 Sci. Bull. 64 301
 Google Scholar
Google Scholar
[29] Wang Y, Miao M, Lv J, Zhu L, Yin K, Liu H, Ma Y 2012 J. Chem. Phys. 137 224108
 Google Scholar
Google Scholar
[30] Hafner J 2008 J. Comput. Chem. 29 2044
 Google Scholar
Google Scholar
[31] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[32] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[33] Sanna S, Schmidt W G, Frauenheim T, Gerstmann U 2009 Phys. Rev. B 80 104120
 Google Scholar
Google Scholar
[34] Xiao Y, Ju M, Yuan H K, Yeung Y Y 2021 J. Phys. Chem. C 125 18015
 Google Scholar
Google Scholar
[35] Togo A, Tanaka I 2015 Scr. Mater. 108 1
 Google Scholar
Google Scholar
[36] Phenicie C M, Stevenson P, Welinski S, Rose B C, Asfaw A T, Cava R J, Lyon S A, de Leon N P, Thompson J D 2019 Nano Lett. 19 8928
 Google Scholar
Google Scholar
[37] Mills A, Hunte S L 1997 J. Photochem. Photobiol. A 108 1
 Google Scholar
Google Scholar
[38] Yang J, Hu Y, Jin C, Zhuge L, Wu X 2017 Thin Solid Films 637 9
 Google Scholar
Google Scholar
[39] Pan L, Xiao Y, Kuang X Y, Ju M 2021 Mater. Chem. Phys. 257 123824
 Google Scholar
Google Scholar
[40] Savin A, Nesper R, Wengert S, Fässler T F 1997 Angew. Chem. Int. Ed. Engl. 36 1808
 Google Scholar
Google Scholar
[41] Lu T, Chen F 2011 Acta Phys. Chim. Sin. 27 2786
 Google Scholar
Google Scholar
[42] Fuentealba P, Chamorro E, Santos J C 2007 Theoretical Aspects of Chemical Reactivity 19 57
Catalog
Metrics
- Abstract views: 7264
- PDF Downloads: 152
- Cited By: 0















 DownLoad:
DownLoad: