-
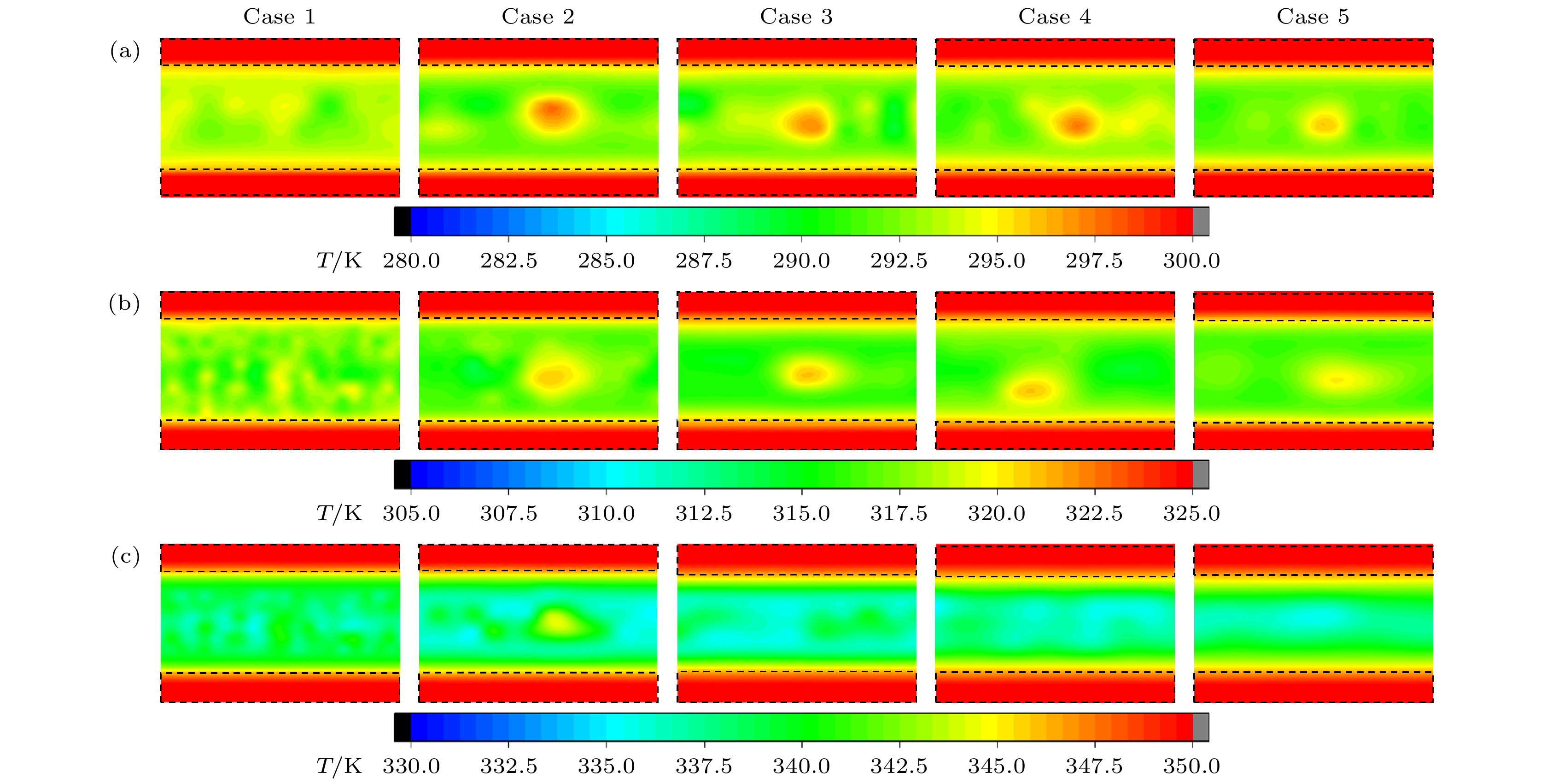
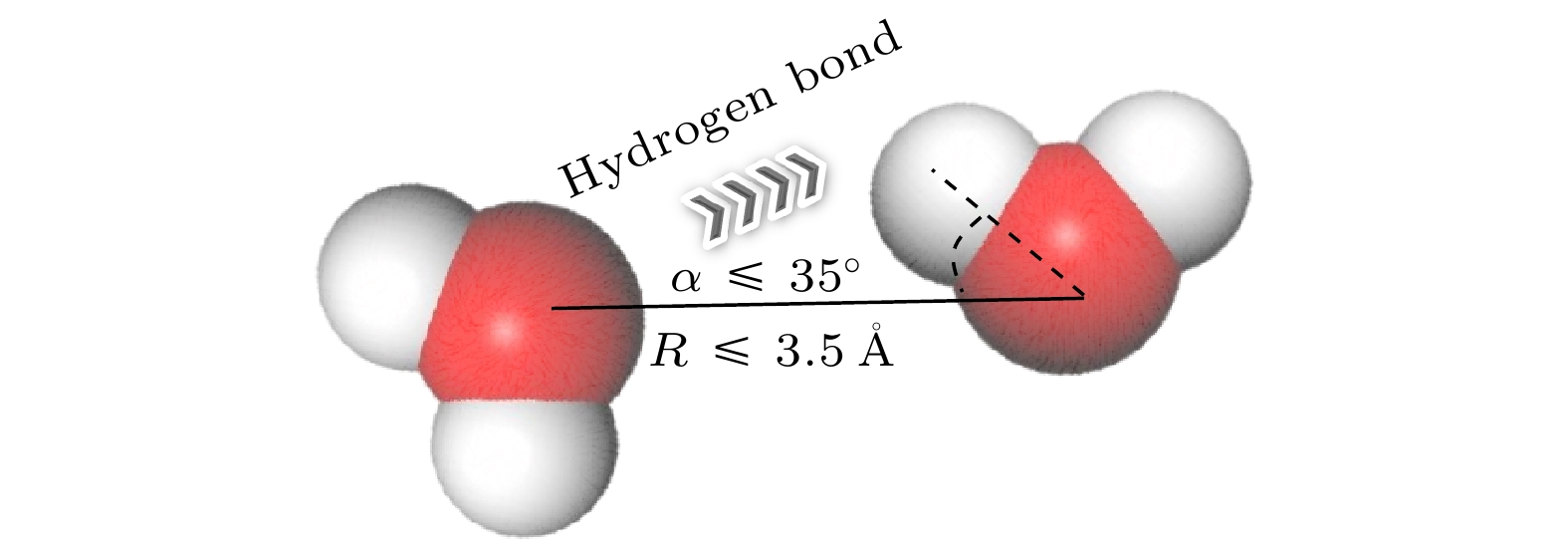
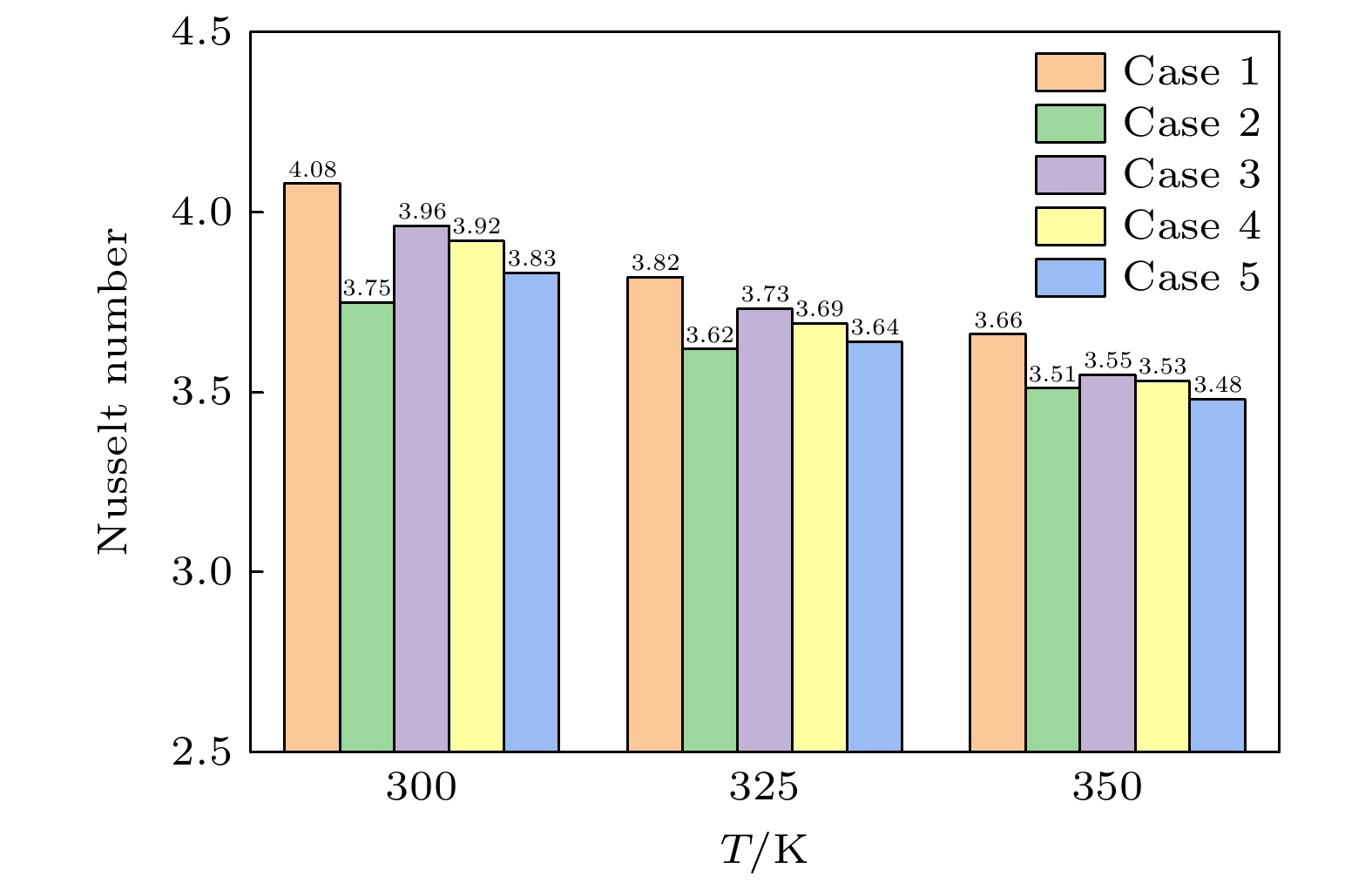
随着电子元件高性能化和小型化的发展, 纳米通道内工质的流动传热问题受到了更多的关注. 本文采用分子动力学模拟方法, 模拟了300, 325, 350 K的纳米通道中流体的流动传热情况, 工质为水, 水中不凝性气体用氩气代替. 结果表明: 流动过程中, 氩原子形成高势能团簇, 随着温度升高, 流体势能上升, 团簇逐渐减小或消失; 少量气体原子能够促进流动, 而较多氩气会导致通道中心区域形成较大气体团簇而阻碍流动, 同时, 被加热的工质能显著减小流动阻力系数; 近壁面区域流体温度高于中心区域, 团簇内部原子活动更加剧烈, 平均分子动能更大, 温度更高; 水的氢键结构可以促进纳米通道内的传热, 氩原子会影响氢键数量, 高温会破坏水分子形成的氢键网络, 使努塞尔数下降. 本研究分析了不凝性气体影响下微通道内水分子流动传热的机理, 为电子设备的强化传热提供了理论指导.With the development of higher performance and miniaturization of electronic components, the flow heat transfer of working fluids in nanochannels has received more attention. To elucidate this phenomenon, molecular dynamics simulations are used to simulate the behaviors of fluids within nanochannels at temperatures of 300 K, 325 K, and 350 K. Water serves as a flow medium, with argon substituted for any non-condensable gases. In the flow process, argon atoms aggregate into clusters that are characterized by high potential energy. As the temperature rises, the concomitant increases in the fluid’s potential energy, which leads to the gradual diminution or complete dissipation of these clusters. A minor presence of gas atoms can facilitate fluid movement; however, an excess of argon promotes the formation of larger gaseous clusters in the central region of the channel, thereby impeding fluid flow. Concurrently, the application of heat to the fluid appreciably diminishes the coefficient of flow resistance. The temperature of the fluid in the near-wall region exceeds that of the central area. In the clusters, the atoms exhibit heightened activity, leading to an increase in the average molecular kinetic energy and a concomitant rise in temperature. The inherent hydrogen-bonding structure of water enhances heat transfer within the nanochannels. Argon atoms exert an influence on the number of hydrogen bonds, and rising temperatures disrupts the hydrogen-bond network established by water molecules, ultimately leading to a decrease of the Nusselt number. This investigation offers insights into the heat transfer dynamics of water molecular flow within microchannels under the perturbation of non-condensable gases, thereby furnishing theoretical guidance for enhancing heat transfer within electronic devices.
-
Keywords:
- molecular dynamics /
- heat transfer /
- cluster /
- nanochannel
[1] Tang J Q, Liu Y, Huang B L, Xu D Y 2022 Int. J. Therm. Sci. 177 107573
 Google Scholar
Google Scholar
[2] Fathi M, Heyhat M M, Targhi M Z, Bigham S 2023 Int. Commun. Heat Mass Transf. 146 106868
 Google Scholar
Google Scholar
[3] Joseph P, Tabeling P 2005 Phys. Rev. E 71 035303
 Google Scholar
Google Scholar
[4] 苗瑞灿, 张石重, 陈占秀, 杨历 2020 化工进展 39 1641
 Google Scholar
Google Scholar
Miao R C, Zhang S Z, Chen Z X, Yang L 2020 Chem. Ind. Eng. Prog. 39 1641
 Google Scholar
Google Scholar
[5] 梅涛, 陈占秀, 杨历, 王坤, 苗瑞灿 2019 68 094701
 Google Scholar
Google Scholar
Mei T, Chen Z X, Yang L, Wang K, Miao R C 2019 Acta Phys. Sin. 68 094701
 Google Scholar
Google Scholar
[6] Shi L, Hu C Z, Bai M L, Lv J Z 2019 Comput. Mater. Sci. 170 109127
 Google Scholar
Google Scholar
[7] Voronov R S, Papavassiliou D V, Lee L L 2007 Chem. Phys. Lett. 441 273
 Google Scholar
Google Scholar
[8] Zhang C B, Chen Y P 2014 Chem. Eng. Process. 85 203
 Google Scholar
Google Scholar
[9] Priezjev N V 2007 Phys. Rev. E 75 051605
 Google Scholar
Google Scholar
[10] Semiromi D T, Azimian A R 2010 Heat Mass Transfer. 46 791
 Google Scholar
Google Scholar
[11] Markvoort A J, Hilbers P A J, Nedea S V 2005 Phys. Rev. E 71 066702
 Google Scholar
Google Scholar
[12] Zhou W B, Han D M, Xia G D 2022 Appl. Surf. Sci. 591 153155
 Google Scholar
Google Scholar
[13] Bao L Y, Priezjev N V, Hu H B, Luo K 2017 Phys. Rev. E 96 033110
 Google Scholar
Google Scholar
[14] Ge S, Gu Y W, Chen M 2015 Mol. Phys. 113 703
 Google Scholar
Google Scholar
[15] Yao S T, Wang J S, Jin S F, Tan F G, Chen S P 2023 J. Mol. Liq. 371 121105
 Google Scholar
Google Scholar
[16] Alkhwaji A, Elbahloul S, Abdullah M Z, Bakar K F B A 2021 J. Mol. Liq. 324 114703
 Google Scholar
Google Scholar
[17] Hong S N, Choe S H, Jong U G, Pak M S, Yu C J 2019 Fluid Phase Equilib. 487 45
 Google Scholar
Google Scholar
[18] Xi B H, Zhao T, Gao Q W, Wei Z X, Zhao S L 2022 Chem. Eng. Sci. 254 117618
 Google Scholar
Google Scholar
[19] 张石重, 陈占秀, 杨历, 苗瑞灿, 张子剑 2020 化工进展 39 3892
 Google Scholar
Google Scholar
Zhang S Z, Chen Z X, Yang L, Miao R C, Zhang Z J 2020 Chem. Ind. Eng. Prog. 39 3892
 Google Scholar
Google Scholar
[20] Chen L, Wang S Y, Xiang X, Tao W Q 2020 Comput. Mater. Sci. 171 109223
 Google Scholar
Google Scholar
[21] Wu N N, Zeng L C, Fu T, Wang Z H, Lu C 2020 Int. J. Heat Mass Transf. 147 118905
 Google Scholar
Google Scholar
[22] Stukowski A 2010 Modell. Simul. Mat. Sci. Eng. 18 015012
 Google Scholar
Google Scholar
[23] Yao S T, Wang J S, Liu X L 2021 Int. J. Heat Mass Transf. 176 121441
 Google Scholar
Google Scholar
[24] Song Z, Shang X S, Cui Z, Liu Y, Cao Q 2023 Appl. Therm. Eng. 218 119362
 Google Scholar
Google Scholar
[25] 梅涛, 陈占秀, 杨历, 朱洪漫, 苗瑞灿 2020 69 224701
 Google Scholar
Google Scholar
Mei T, Chen Z X, Yang L, Zhu H M, Miao R C 2020 Acta Phys. Sin. 69 224701
 Google Scholar
Google Scholar
[26] Tao J B, Song X Y, Chen W, Zhao S L, Liu H L 2020 Mol. Simul. 46 699
 Google Scholar
Google Scholar
[27] Motlagh M B, Kalteh M 2020 J. Mol. Liq. 318 114028
 Google Scholar
Google Scholar
-
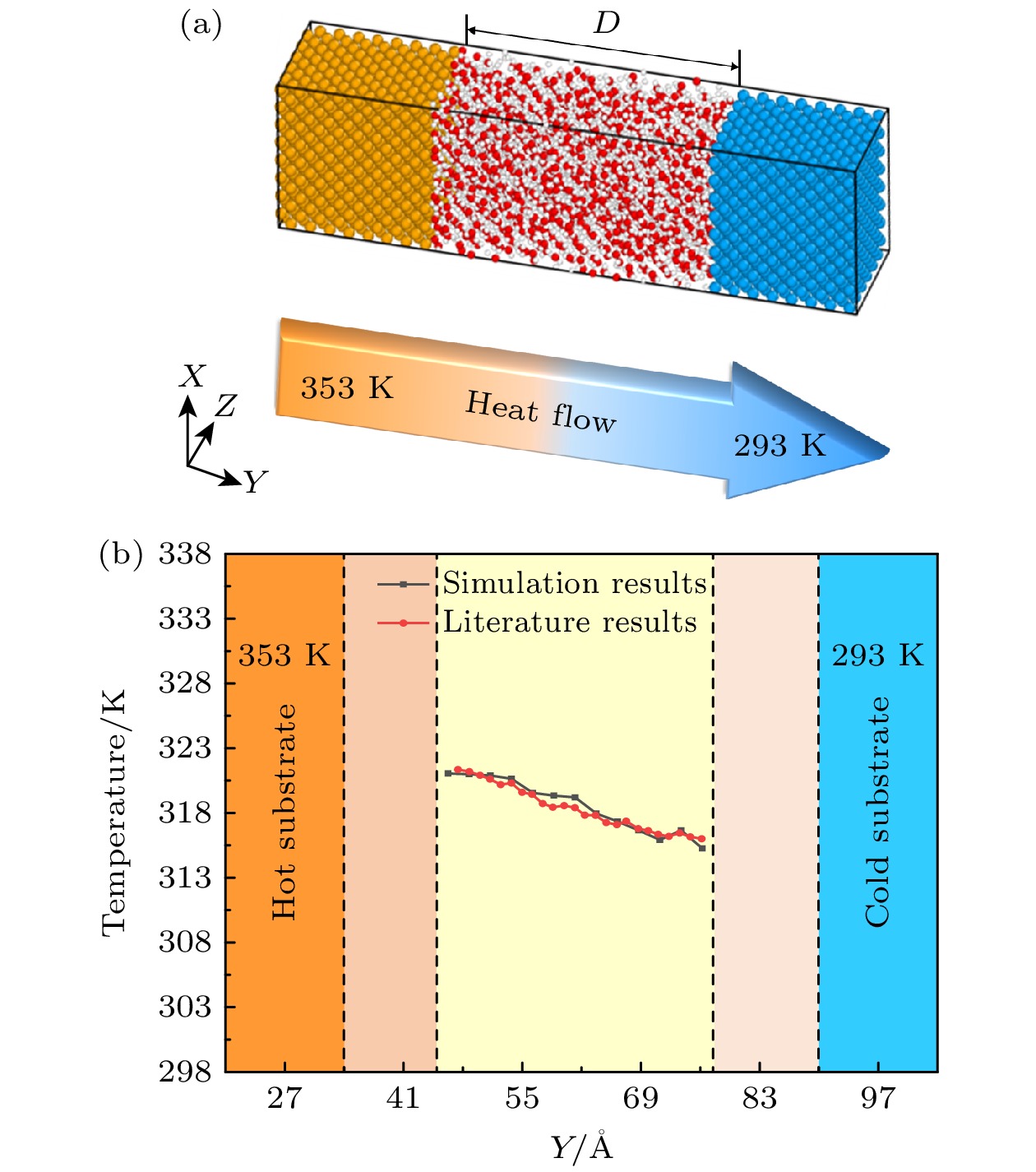
图 4 流体的数密度分布 (a) T0 = 300 K, H2O; (b) T0 = 300 K, Ar; (c) T0 = 325 K, H2O; (d) T0 = 325 K, Ar; (e) T0 = 350 K, H2O; (f) T0 = 350 K, Ar
Fig. 4. Number density distribution of fluids: (a) T0 = 300 K, H2O; (b) T0 = 300 K, Ar; (c) T0 = 325 K, H2O; (d) T0 = 325 K, Ar; (e) T0 = 350 K, H2O; (f) T0 = 350 K, Ar.
表 1 流道内的粒子数量
Table 1. Number of particles in the channel.
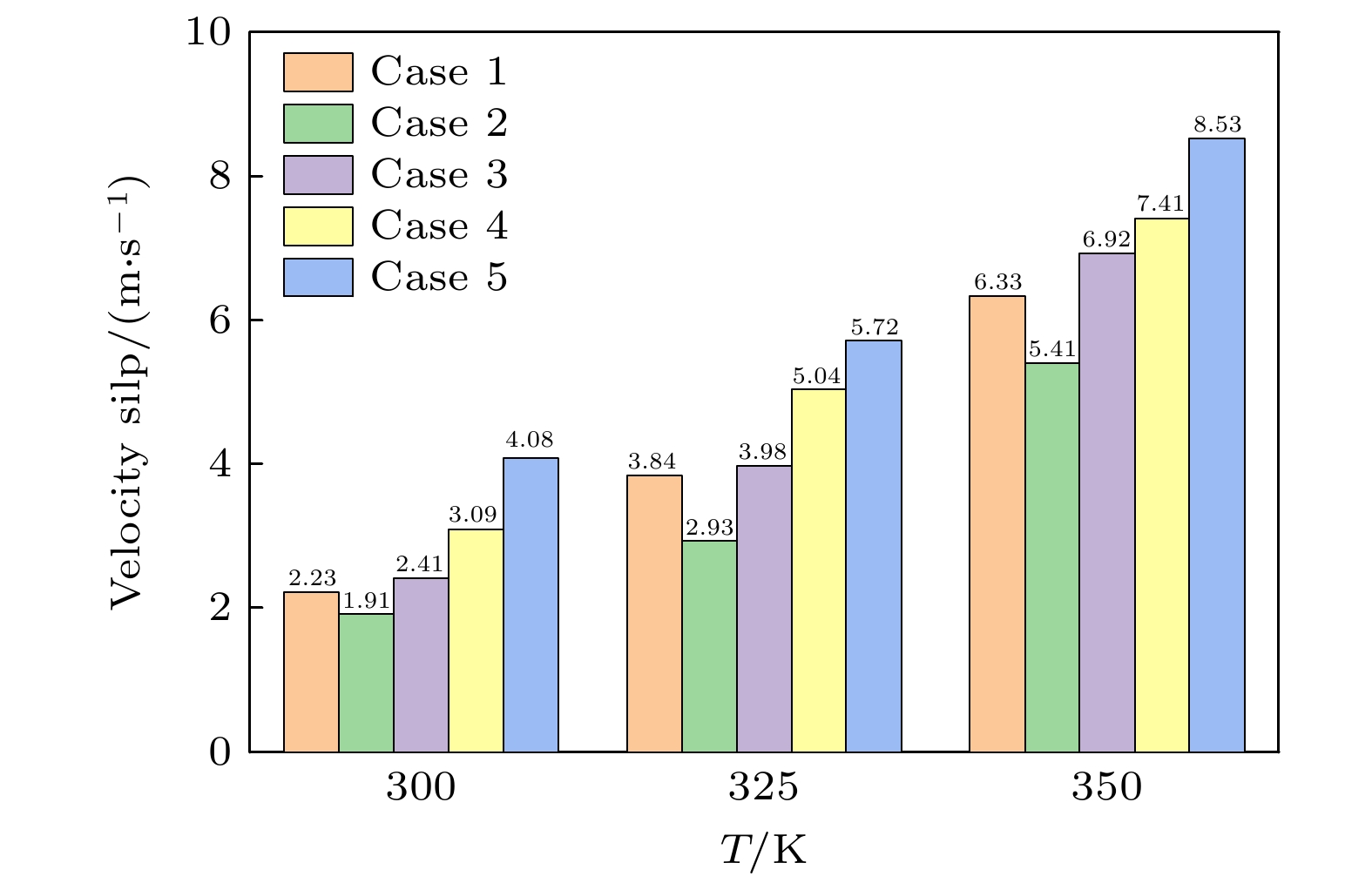
${N_{{{\text{H}}_{2}}{\text{O}}}}$ NAr Case 1 2600 0 Case 2 2200 600 Case 3 2400 400 Case 4 2400 300 Case 5 2400 120 表 2 各原子之间的相互作用势能参数及尺寸参数
Table 2. Interaction potential energy parameters and size parameters of each atom.
原子种类 势能参数 ε/(kcal·mol–1) 尺寸参数 σ/nm O-O 0.1554 0.3166 H-H 0 0 Ar-Ar 0.2392 0.3040 Cu-Cu 0.2379 0.2340 -
[1] Tang J Q, Liu Y, Huang B L, Xu D Y 2022 Int. J. Therm. Sci. 177 107573
 Google Scholar
Google Scholar
[2] Fathi M, Heyhat M M, Targhi M Z, Bigham S 2023 Int. Commun. Heat Mass Transf. 146 106868
 Google Scholar
Google Scholar
[3] Joseph P, Tabeling P 2005 Phys. Rev. E 71 035303
 Google Scholar
Google Scholar
[4] 苗瑞灿, 张石重, 陈占秀, 杨历 2020 化工进展 39 1641
 Google Scholar
Google Scholar
Miao R C, Zhang S Z, Chen Z X, Yang L 2020 Chem. Ind. Eng. Prog. 39 1641
 Google Scholar
Google Scholar
[5] 梅涛, 陈占秀, 杨历, 王坤, 苗瑞灿 2019 68 094701
 Google Scholar
Google Scholar
Mei T, Chen Z X, Yang L, Wang K, Miao R C 2019 Acta Phys. Sin. 68 094701
 Google Scholar
Google Scholar
[6] Shi L, Hu C Z, Bai M L, Lv J Z 2019 Comput. Mater. Sci. 170 109127
 Google Scholar
Google Scholar
[7] Voronov R S, Papavassiliou D V, Lee L L 2007 Chem. Phys. Lett. 441 273
 Google Scholar
Google Scholar
[8] Zhang C B, Chen Y P 2014 Chem. Eng. Process. 85 203
 Google Scholar
Google Scholar
[9] Priezjev N V 2007 Phys. Rev. E 75 051605
 Google Scholar
Google Scholar
[10] Semiromi D T, Azimian A R 2010 Heat Mass Transfer. 46 791
 Google Scholar
Google Scholar
[11] Markvoort A J, Hilbers P A J, Nedea S V 2005 Phys. Rev. E 71 066702
 Google Scholar
Google Scholar
[12] Zhou W B, Han D M, Xia G D 2022 Appl. Surf. Sci. 591 153155
 Google Scholar
Google Scholar
[13] Bao L Y, Priezjev N V, Hu H B, Luo K 2017 Phys. Rev. E 96 033110
 Google Scholar
Google Scholar
[14] Ge S, Gu Y W, Chen M 2015 Mol. Phys. 113 703
 Google Scholar
Google Scholar
[15] Yao S T, Wang J S, Jin S F, Tan F G, Chen S P 2023 J. Mol. Liq. 371 121105
 Google Scholar
Google Scholar
[16] Alkhwaji A, Elbahloul S, Abdullah M Z, Bakar K F B A 2021 J. Mol. Liq. 324 114703
 Google Scholar
Google Scholar
[17] Hong S N, Choe S H, Jong U G, Pak M S, Yu C J 2019 Fluid Phase Equilib. 487 45
 Google Scholar
Google Scholar
[18] Xi B H, Zhao T, Gao Q W, Wei Z X, Zhao S L 2022 Chem. Eng. Sci. 254 117618
 Google Scholar
Google Scholar
[19] 张石重, 陈占秀, 杨历, 苗瑞灿, 张子剑 2020 化工进展 39 3892
 Google Scholar
Google Scholar
Zhang S Z, Chen Z X, Yang L, Miao R C, Zhang Z J 2020 Chem. Ind. Eng. Prog. 39 3892
 Google Scholar
Google Scholar
[20] Chen L, Wang S Y, Xiang X, Tao W Q 2020 Comput. Mater. Sci. 171 109223
 Google Scholar
Google Scholar
[21] Wu N N, Zeng L C, Fu T, Wang Z H, Lu C 2020 Int. J. Heat Mass Transf. 147 118905
 Google Scholar
Google Scholar
[22] Stukowski A 2010 Modell. Simul. Mat. Sci. Eng. 18 015012
 Google Scholar
Google Scholar
[23] Yao S T, Wang J S, Liu X L 2021 Int. J. Heat Mass Transf. 176 121441
 Google Scholar
Google Scholar
[24] Song Z, Shang X S, Cui Z, Liu Y, Cao Q 2023 Appl. Therm. Eng. 218 119362
 Google Scholar
Google Scholar
[25] 梅涛, 陈占秀, 杨历, 朱洪漫, 苗瑞灿 2020 69 224701
 Google Scholar
Google Scholar
Mei T, Chen Z X, Yang L, Zhu H M, Miao R C 2020 Acta Phys. Sin. 69 224701
 Google Scholar
Google Scholar
[26] Tao J B, Song X Y, Chen W, Zhao S L, Liu H L 2020 Mol. Simul. 46 699
 Google Scholar
Google Scholar
[27] Motlagh M B, Kalteh M 2020 J. Mol. Liq. 318 114028
 Google Scholar
Google Scholar
计量
- 文章访问数: 2772
- PDF下载量: 50
- 被引次数: 0














 下载:
下载: