-
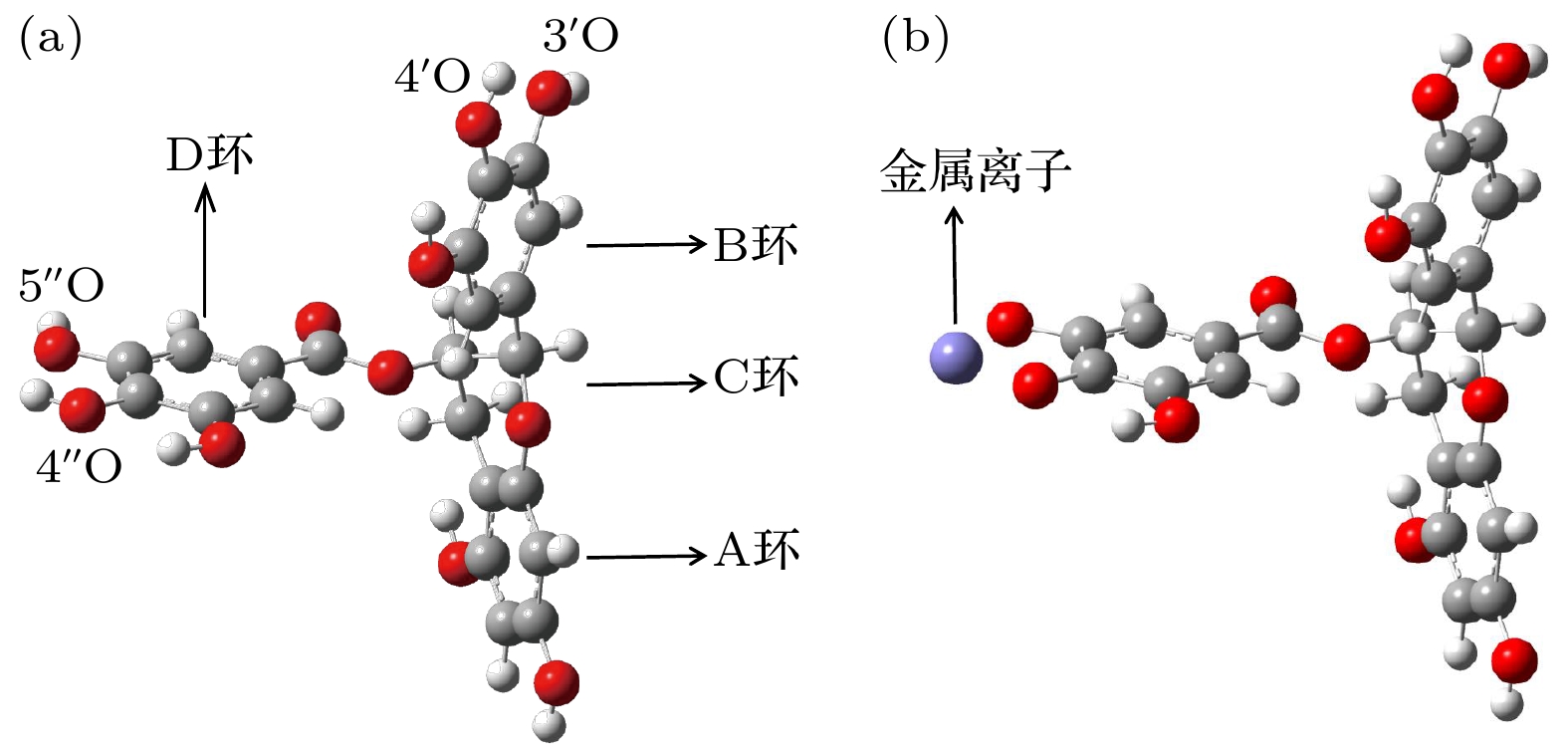
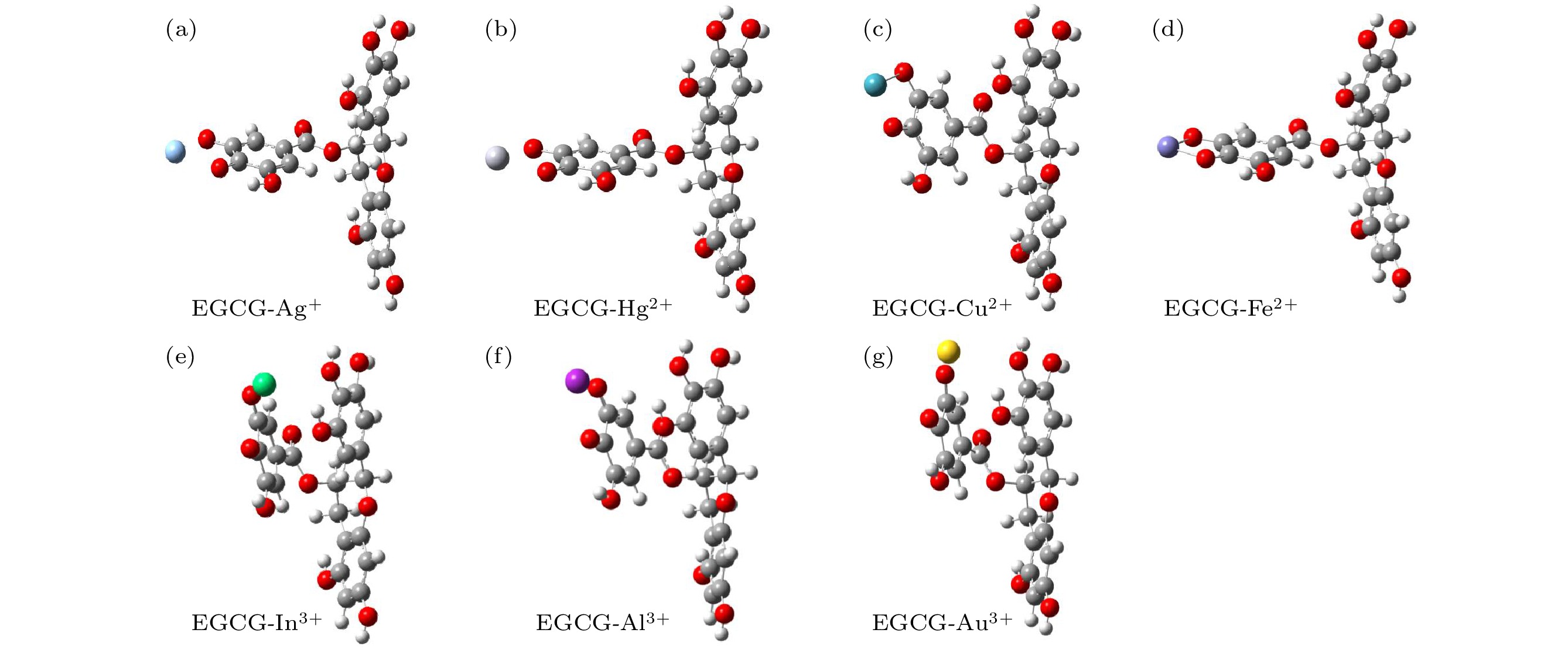
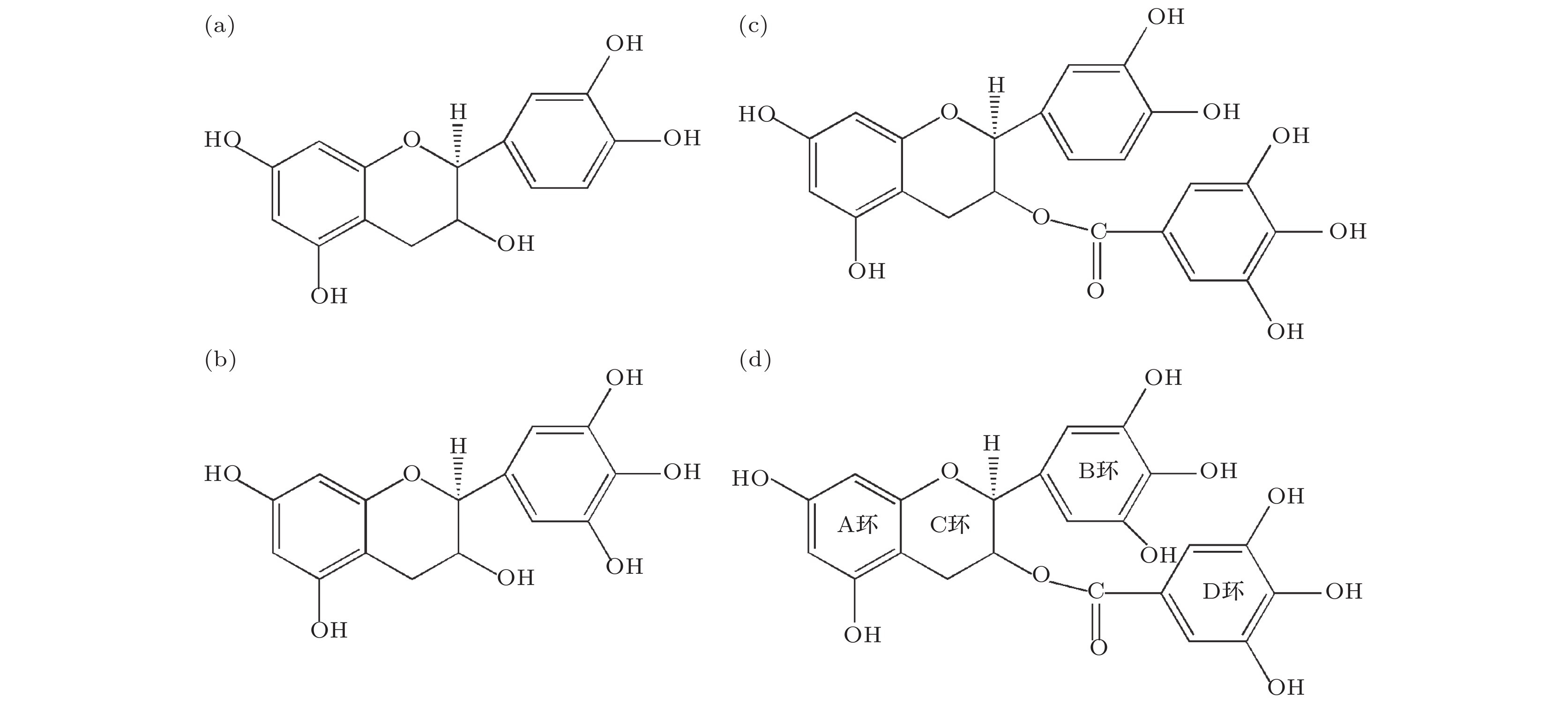
Persimmon tannin has excellent adsorption efficiency of heavy metal ions, and epigallocatechin gallate (EGCG) is the key structural monomer of persimmon tannin to play its active role. In order to analyze the nature of the interaction between persimmon tannin and metal ions, in this paper the density functional theory (DFT) is used to calculate and analyze the interactions between EGCG and metal ions (Ag+, Hg2+, Cu2+, Fe2+, In3+, Al3+, Au3+), from the respects of EGCG-metal complex configuration, Mayer bond order, natural population analysis, binding energy, and weak interaction. In this paper, the B3LYP combined with DFT-D3 dispersion correction method is mainly used. For metal atoms, the Lanl2dz basis set is adopted. For H, C and O atoms, the 6-311G (d, p) basis set is adopted for optimizing the structure, and the more accurate 6-311+G (d, p) basis set is selected for calculating the single point energy. At the same time, the study adds the SMD solvation model with water as the solvent. All calculations are done by using the Gaussian 09 package. The method of reduced density gradient function is used to study the weak interactions between EGCG and metal ions. The results of research show that EGCG-Fe2+ complex is adsorbed mainly by chelating bond. However, the EGCG adsorbs mainly Ag+, Hg2+ ions through electrostatic attraction. The configurations of the complexes show that In3+, Al3+ and Au3+ ions with EGCG form unique “luminal structure” metal complexes, so there is not only electrostatic attraction, but also aromatic ring stacking between these three metal ions and D ring 4"O, 5"O. The calculated Mayer bond order indicates that the bond order of the composite bond is formed by Fe2+ ion and the EGCG is the largest in the seven metal complexes, and the bond order is formed by In3+ ion, and EGCG is smallest. The compound of Cu2+ ion and EGCG have chelation, electrostatic attraction and aromatic ring stacking. By observing the binding energy, it can be found that the more charges the metal ions have, the easier the charge transfer will be and the stronger the electrostatic attraction of EGCG may be. These results will provide enlightenment for further studying the mechanism of persimmon tannin's adsorption of metal ions.
-
Keywords:
- persimmon tannin /
- metal ion /
- absorb /
- density functional theory
[1] Matheus J R V, Andrade C J D, Miyahira R F, Fai A E C 2020 Food Rev. Int.
 Google Scholar
Google Scholar
[2] Elhabiri M, Carrer C, Marmolle F, Traboulsi H 2007 Inorg. Chim. Acta 360 353
 Google Scholar
Google Scholar
[3] Chen Y M, Wang M K, Huang P M 2006 J. Agric. Food Chem. 54 212
 Google Scholar
Google Scholar
[4] Li X J, Wang Z M, Liang H J, Ning J L, Li G Y, Zhou Z D 2017 Environ. Technol. 40 112
 Google Scholar
Google Scholar
[5] Wang Z M, Gao M M, Li X J, Ning J L, Zhou Z D, Li G Y 2020 Mater. Sci. Eng., C 108 110196
 Google Scholar
Google Scholar
[6] Nakajima A, Sakaguchi T 1993 J. Chem. Technol. Biotechnol. 57 321
 Google Scholar
Google Scholar
[7] Gurung M, Adhikari B B, Kawakita H, Ohto K, Inoue K, Alam S 2012 Ind. Eng. Chem. Res. 51 11901
 Google Scholar
Google Scholar
[8] Zhou Z D, Liu F L, Huang Y, Wang Z M, Li G Y 2015 Int. J. Biol. Macromol. 77 336
 Google Scholar
Google Scholar
[9] Gurung M, Adhikari B B, Morisada S, Kawakita H, Ohto K, Inoue K, Alam S 2013 Bioresource Technol. 129 108
 Google Scholar
Google Scholar
[10] Yi Q, Fan R, Xie F, Zhang Q, Luo Z 2016 J. Taiwan Inst. Chem. Eng. 61 299
 Google Scholar
Google Scholar
[11] Li C M, Leverence R, Trombley J D, Xu S, Yang J, Tian Y, Reed J D 2010 J. Agric. Food Chem. 58 9033
 Google Scholar
Google Scholar
[12] 江腾, 马万福, 谢楠, 周平 2011 物理化学学报 27 2291
 Google Scholar
Google Scholar
Jiang T, Ma W F, Xie N, Zhou P 2011 Acta Phys-Chim. Sin. 27 2291
 Google Scholar
Google Scholar
[13] 王晓巍, 蒋刚, 杜际广 2011 物理化学学报 27 309
 Google Scholar
Google Scholar
Wang X W, Jiang G, Du J G 2011 Acta Phys.-Chim. Sin. 27 309
 Google Scholar
Google Scholar
[14] Kohn W, Sham L J 1965 Phys. Rev. 140 A1133
 Google Scholar
Google Scholar
[15] Becke A D 1993 J. Chem. Phys. 98 5648
 Google Scholar
Google Scholar
[16] Malgorzata B, Pawel P, Giovanni S, Julien B, Vincenzo B 2010 J. Chem. Theory Comput. 6 2115
 Google Scholar
Google Scholar
[17] Jonathon W, Matthew G, Jeffrey B N, Martin H 2015 J. Chem. Theory Comput. 11 1481
 Google Scholar
Google Scholar
[18] Becke A D 1988 Phys. Rev. A 38 3098
 Google Scholar
Google Scholar
[19] Lee C, Yang W, Parr R G 1988 Phys. Rev. B 37 785
 Google Scholar
Google Scholar
[20] Bougherara H, Kadri R, Kadri M, Yekhlef M, Boumaza A 2021 J. Mol. Struct. 1223 128855
 Google Scholar
Google Scholar
[21] Viji A, Revathi B, Balachandran V, Babiyana S, Narayana B, Salian V V 2020 Chem.l Data Collections 30 100585
 Google Scholar
Google Scholar
[22] Goerigk L, Grimme S 2011 Phys. Chem. Chem. Phys. 13 6670
 Google Scholar
Google Scholar
[23] Frisch M J, Trucks G W, Schlegel H B, et al. 2009 Gaussian 09 (Rev. A.02). (Gaussian: Inc., Wallingford CT)
[24] Johnson E R, Kernan S, Mori-Sanchez P, Contreras-Garcia J, Cohen A J, Yang W T 2010 J. Am. Chem. Soc. 132 6498
 Google Scholar
Google Scholar
[25] 施斌, 袁荔, 唐天宇, 陆利敏, 赵先豪, 魏晓楠, 唐延林 2021 70 053102
 Google Scholar
Google Scholar
Shi B, Yuan L, Tang T Y, Lu L M, Zhao X H, W X N, Tang Y L 2021 Acta Phys. Sin. 70 053102
 Google Scholar
Google Scholar
[26] Navarro R E, Santacruz H, Inoue M 2005 J. Inorg. Biochem. 99 584
 Google Scholar
Google Scholar
[27] Inoue M B, Inoue M, Fernando Q, Valcic S, Timmermann B N 2002 J. Inorg. Biochem. 88 7
 Google Scholar
Google Scholar
[28] Esparza I, Salinas I, Santamaria C, Garcia-Mina J M, Fernandez J M 2005 Anal. Chim. Acta 543 267
 Google Scholar
Google Scholar
[29] Lu T, Chen F W 2013 J. Phys. Chem. A. 117 3100
 Google Scholar
Google Scholar
[30] 卢天, 陈飞武 2012 物理化学学报 28 1
 Google Scholar
Google Scholar
Lu T, Chen F W 2012 Acta Phys.-Chim. Sin. 28 1
 Google Scholar
Google Scholar
[31] Lu T, Chen F W 2012 J. Comput. Chem. 33 580
 Google Scholar
Google Scholar
[32] Humphrey W, Dalke A, Schulten K 1996 J. Mol. Graphics. 14 33
 Google Scholar
Google Scholar
-
表 1 EGCG-金属复合物的复合键键长
Table 1. Composite bond lengths of the EGCG-metal complexes.
EGCG-金属复合物 复合键 键长/Å ECGC-Ag+ 4"O—Ag 2.363 5"O—Ag 2.302 EGCG-Hg2+ 4"O—Hg 2.366 5"O—Hg 2.333 EGCG-Cu2+ 4"O—Cu 1.941 5"O—Cu 1.916 EGCG-Fe2+ 4"O—Fe 1.926 5"O—Fe 1.893 EGCG-In3+ 4"O—In 2.929 5"O—In 2.683 EGCG-Al3+ 4"O—Al 2.418 5"O—Al 2.008 EGCG-Au3+ 4"O—Au 2.979 5"O—Au 2.194 表 2 EGCG-金属复合物中复合键的Mayer键级
Table 2. The Mayer bond orders of composite bond in the EGCG-metal complexes.
EGCG-金属复合物 复合键 Mayer键级 ECGC-Ag+ 4"O—Ag 0.3462 5"O—Ag 0.4304 EGCG-Hg2+ 4"O—Hg 0.3870 5"O—Hg 0.4697 EGCG-Cu2+ 4"O—Cu 0.5089 5"O—Cu 0.5458 EGCG-Fe2+ 4"O—Fe 0.5940 5"O—Fe 0.7480 EGCG-In3+ 4"O—In 0.0742 5"O—In 0.1183 EGCG-Al3+ 4"O—Al 0.1635 5"O—Al 0.3022 EGCG-Au3+ 4"O—Au 0.1058 5"O—Au 0.4331 表 3 EGCG-金属复合物中复合原子的自然布居分析
Table 3. Natural population analysis of composite atoms in the EGCG-metal complexes.
EGCG-金属复合物 复合原子 自然电荷分布 ECGC-Ag+ Ag 0.7803 4"O –0.8837 5"O –0.9144 EGCG-Hg2+ Hg 1.7086 4"O –0.8860 5"O –0.9045 EGCG-Cu2+ Cu 1.4836 4"O –0.8479 5"O –0.8421 EGCG-Fe2+ Fe 1.4342 4"O –0.8121 5"O –0.8417 EGCG-In3+ In 1.0761 4"O –0.5587 5"O –0.6041 EGCG-Al3+ Al 0.8686 4"O –0.5978 5"O –0.7942 EGCG-Au3+ Au 0.8679 4"O –0.5229 5"O –0.6244 -
[1] Matheus J R V, Andrade C J D, Miyahira R F, Fai A E C 2020 Food Rev. Int.
 Google Scholar
Google Scholar
[2] Elhabiri M, Carrer C, Marmolle F, Traboulsi H 2007 Inorg. Chim. Acta 360 353
 Google Scholar
Google Scholar
[3] Chen Y M, Wang M K, Huang P M 2006 J. Agric. Food Chem. 54 212
 Google Scholar
Google Scholar
[4] Li X J, Wang Z M, Liang H J, Ning J L, Li G Y, Zhou Z D 2017 Environ. Technol. 40 112
 Google Scholar
Google Scholar
[5] Wang Z M, Gao M M, Li X J, Ning J L, Zhou Z D, Li G Y 2020 Mater. Sci. Eng., C 108 110196
 Google Scholar
Google Scholar
[6] Nakajima A, Sakaguchi T 1993 J. Chem. Technol. Biotechnol. 57 321
 Google Scholar
Google Scholar
[7] Gurung M, Adhikari B B, Kawakita H, Ohto K, Inoue K, Alam S 2012 Ind. Eng. Chem. Res. 51 11901
 Google Scholar
Google Scholar
[8] Zhou Z D, Liu F L, Huang Y, Wang Z M, Li G Y 2015 Int. J. Biol. Macromol. 77 336
 Google Scholar
Google Scholar
[9] Gurung M, Adhikari B B, Morisada S, Kawakita H, Ohto K, Inoue K, Alam S 2013 Bioresource Technol. 129 108
 Google Scholar
Google Scholar
[10] Yi Q, Fan R, Xie F, Zhang Q, Luo Z 2016 J. Taiwan Inst. Chem. Eng. 61 299
 Google Scholar
Google Scholar
[11] Li C M, Leverence R, Trombley J D, Xu S, Yang J, Tian Y, Reed J D 2010 J. Agric. Food Chem. 58 9033
 Google Scholar
Google Scholar
[12] 江腾, 马万福, 谢楠, 周平 2011 物理化学学报 27 2291
 Google Scholar
Google Scholar
Jiang T, Ma W F, Xie N, Zhou P 2011 Acta Phys-Chim. Sin. 27 2291
 Google Scholar
Google Scholar
[13] 王晓巍, 蒋刚, 杜际广 2011 物理化学学报 27 309
 Google Scholar
Google Scholar
Wang X W, Jiang G, Du J G 2011 Acta Phys.-Chim. Sin. 27 309
 Google Scholar
Google Scholar
[14] Kohn W, Sham L J 1965 Phys. Rev. 140 A1133
 Google Scholar
Google Scholar
[15] Becke A D 1993 J. Chem. Phys. 98 5648
 Google Scholar
Google Scholar
[16] Malgorzata B, Pawel P, Giovanni S, Julien B, Vincenzo B 2010 J. Chem. Theory Comput. 6 2115
 Google Scholar
Google Scholar
[17] Jonathon W, Matthew G, Jeffrey B N, Martin H 2015 J. Chem. Theory Comput. 11 1481
 Google Scholar
Google Scholar
[18] Becke A D 1988 Phys. Rev. A 38 3098
 Google Scholar
Google Scholar
[19] Lee C, Yang W, Parr R G 1988 Phys. Rev. B 37 785
 Google Scholar
Google Scholar
[20] Bougherara H, Kadri R, Kadri M, Yekhlef M, Boumaza A 2021 J. Mol. Struct. 1223 128855
 Google Scholar
Google Scholar
[21] Viji A, Revathi B, Balachandran V, Babiyana S, Narayana B, Salian V V 2020 Chem.l Data Collections 30 100585
 Google Scholar
Google Scholar
[22] Goerigk L, Grimme S 2011 Phys. Chem. Chem. Phys. 13 6670
 Google Scholar
Google Scholar
[23] Frisch M J, Trucks G W, Schlegel H B, et al. 2009 Gaussian 09 (Rev. A.02). (Gaussian: Inc., Wallingford CT)
[24] Johnson E R, Kernan S, Mori-Sanchez P, Contreras-Garcia J, Cohen A J, Yang W T 2010 J. Am. Chem. Soc. 132 6498
 Google Scholar
Google Scholar
[25] 施斌, 袁荔, 唐天宇, 陆利敏, 赵先豪, 魏晓楠, 唐延林 2021 70 053102
 Google Scholar
Google Scholar
Shi B, Yuan L, Tang T Y, Lu L M, Zhao X H, W X N, Tang Y L 2021 Acta Phys. Sin. 70 053102
 Google Scholar
Google Scholar
[26] Navarro R E, Santacruz H, Inoue M 2005 J. Inorg. Biochem. 99 584
 Google Scholar
Google Scholar
[27] Inoue M B, Inoue M, Fernando Q, Valcic S, Timmermann B N 2002 J. Inorg. Biochem. 88 7
 Google Scholar
Google Scholar
[28] Esparza I, Salinas I, Santamaria C, Garcia-Mina J M, Fernandez J M 2005 Anal. Chim. Acta 543 267
 Google Scholar
Google Scholar
[29] Lu T, Chen F W 2013 J. Phys. Chem. A. 117 3100
 Google Scholar
Google Scholar
[30] 卢天, 陈飞武 2012 物理化学学报 28 1
 Google Scholar
Google Scholar
Lu T, Chen F W 2012 Acta Phys.-Chim. Sin. 28 1
 Google Scholar
Google Scholar
[31] Lu T, Chen F W 2012 J. Comput. Chem. 33 580
 Google Scholar
Google Scholar
[32] Humphrey W, Dalke A, Schulten K 1996 J. Mol. Graphics. 14 33
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 7856
- PDF Downloads: 129
- Cited By: 0














 DownLoad:
DownLoad: