-
低维材料表面的有毒气体吸附一直是分子传感器领域的研究热点. 最近的新型正交相二维BN单层半导体材料引起了学者的广泛关注. 基于第一性原理计算, 本文研究了有毒气体分子(CO, H2S, NH3, NO, NO2和SO2)在二维正交相BN单层表面的吸附特性. 吸附能计算表明, 正交相BN材料对所有气体的吸附均是能量有利的放热过程.NO2和NH3为化学吸附, 其他体系均为物理吸附, 且NO吸附后体系具有自旋极化的电子能带结构. 态密度计算结果显示, NO2和SO2的吸附对费米能级附近的电子占据有显著影响.NO2吸附后, 体系的功函数和电子结构特性均明显改变, 表明BN单层半导体材料对NO2分子具有良好的吸附选择性和探测灵敏度. 采用第一性原理结合非平衡格林函数方法计算了纳米电子器件的输运性能. 在1 V偏压下, NO2分子的吸附可使体系电流增大到6500 nA, 显著增强了BN单层半导体器件的电输运性能, 因此二维BN单层对NO2气体吸附具有优异的探测敏感度. 此外, 轴向应力能够有效调控体系的吸附性能. 研究表明, 正交相BN单层材料对有毒气体具有优异的吸附性能, 在新型有毒气体传感探测器件领域有较大的应用潜力.The adsorption properties of toxic gases on the surface of low-dimensional nanomaterials are a research hot topic and key issue for developing semiconductor sensors to detect toxic gas molecules. Recently, a novel orthorhombic BN monolayer has attracted extensive attention from researchers. Using first principles calculations, we investigate the adsorption properties of typical toxic gas molecules, such as CO, H2S, NH3, NO, NO2, and SO2 molecules, on the surface of two-dimensional (2D) orthorhombic BN monolayer adsorption. The calculated adsorption energy show that the adsorptions of the above six molecules on the surface of BN monolayer are energy-favorable exothermic processes. It is found that NO2 and NH3 molecules are of chemical adsorption, while other systems are of physical adsorption, and NO adsorbing system exhibits a spin-polarized electronic band structure. The calculated density of states reveals that the adsorption of NO molecule and SO2 molecule have significant influences on the electronic structure near the Fermi level. Moreover, the adsorption of the NO2 molecule on the substrate exhibits remarkable variation of the work function, suggesting that the o-BN monolayer possesses excellent selectivity and sensitivity to NO2 molecule. In addition, we use first principles combined with non-equilibrium Green’s function to simulate the electrical transport properties of monolayered o-BN semiconductor based nanodevice with adsorption of typical toxic gas molecules. The I-Vb curve shows that the current through the nanodevice is 6500 nA for the NO2 molecule adsorbing system under 1 V bias voltage. The calculation results reveal that the adsorption of NO2 molecule on the o-BN monolayer can significantly enhance its electrical transport performance, and the o-BN monolayer possesses excellent sensitivity and selectivity to the NO2 gas molecule. The work function and the charge transfer can be effectively manipulated by tensile strain, indicating its potential application in anisotropic electronics. Our results indicate that the o-BN monolayer has excellent adsorption performance to toxic gases, showing its practical application in capturing toxic gas molecules as a gas sensor in future.
-
Keywords:
- adsorption /
- orthorhombic BN /
- first-principles /
- electrical transport /
- gas sensor
[1] Radisavljevic B, Radenovic A, Brivio J, Giacometti V, Kis A 2011 Nat. Nanotechnol. 6 147
 Google Scholar
Google Scholar
[2] Tombros N, Jozsa C, Popinciuc M, Jonkman H T, Wees B 2007 Nature 448 571
 Google Scholar
Google Scholar
[3] Allen M, Tung V, Kaner R 2010 Chem. Rev. 110 132
 Google Scholar
Google Scholar
[4] Wang G Y, Yu J B, Zheng K, Huang Y X, Li X D, Chen X P, Tao L Q 2020 IEEE Electron. Device Lett. 41 1404
 Google Scholar
Google Scholar
[5] Ma S X, Li D J, Rao X J, Xia X F, Su Y, Lu Y F 2020 Adsorption 26 619
 Google Scholar
Google Scholar
[6] Lin L Y, Liu T M, Zhang Y, Sun R, Zeng W, Wang Z C 2016 Ceram. Int. 42 971
 Google Scholar
Google Scholar
[7] Feng C, Qin H B, Yang D G, Zhang G Q 2019 Materials 12 676
 Google Scholar
Google Scholar
[8] Ren J, Kong W, Ni J 2019 Nanoscale Res. Lett. 14 1
 Google Scholar
Google Scholar
[9] Sun X, Yang Q, Meng R S, Tan C J, Liang Q H, Jiang J K, Ye H Y, Chen X P 2017 Appl. Surf. Sci. 404 291
 Google Scholar
Google Scholar
[10] Shahriar R, Hassan O, Alam M K 2022 RSC Adv. 12 16732
 Google Scholar
Google Scholar
[11] 徐强, 段康, 谢浩, 张秦蓉, 梁本权, 彭祯凯, 李卫 2021 70 157101
 Google Scholar
Google Scholar
Xu Q, Duan K, Xie H, Zhang Q R, Liang B Q, Peng Z K, Li W 2021 Acta Phys. Sin. 70 157101
 Google Scholar
Google Scholar
[12] Zhang D Z, Pan W J, Tang M C, Wang D Y, Yu S J, Mi Q, Pan Q N, Hu Y Q 2023 Nano Res. 16 11959
 Google Scholar
Google Scholar
[13] Demirci S, Rad S E, Kazak S, Nezir S, Jahangirov S 2020 Phys. Rev. B 101 125408
 Google Scholar
Google Scholar
[14] Zhao J, Zeng H, Yao G 2021 Phys. Chem. Chem. Phys. 23 3771
 Google Scholar
Google Scholar
[15] Rahimi R, Solimannejad M 2021 Energy Fuels 35 6858
 Google Scholar
Google Scholar
[16] Khossossi N, Luo W, Haman Z, Singh D, Essaoudi I, Ainane A, Ahuja R 2022 Nano Energy 96 107066
 Google Scholar
Google Scholar
[17] Ma D W, Lu Z S, Ju W W, et al. 2012 J. Phys.: Condens. Mat. 24 145501
 Google Scholar
Google Scholar
[18] Zhou Y G, Dong J X, Wang Z G, Xiao H Y, Gao F, Zu X T 2010 Phys. Chem. Chem. Phys. 12 7588
 Google Scholar
Google Scholar
[19] Zhou Y G, Zu X T, Yang P, et al. 2010 J. Phys.: Condens. Mat. 22 465303
 Google Scholar
Google Scholar
[20] Li J, Hu M L, Yu Z Z, Zhong J X, Sun L Z 2012 Chem. Phys. Lett. 532 40
 Google Scholar
Google Scholar
[21] Xu R H, Kong F, Wan J, Guo J Y 2023 Mat. Sci. Semicon. Proc. 161 107446
 Google Scholar
Google Scholar
[22] Paghi A, Mariani S, Barillaro G 2023 Small 19 2206100
 Google Scholar
Google Scholar
[23] Yang S, Jiang C, Wei S H 2017 Appl. Phys. Rev. 4 021304
 Google Scholar
Google Scholar
[24] Kresse G, Hafner J 1993 Phys. Rev. B 47 558
 Google Scholar
Google Scholar
[25] Kresse G, Furthmuller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[26] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[27] Grimme S 2006 J. Comput. Chem. 27 1787
 Google Scholar
Google Scholar
[28] Monkhorst H J, Pack J D 1976 Phys. Rev. B 13 5188
 Google Scholar
Google Scholar
[29] Zhang J N, Ma L, Zhang M, Zhang J M 2020 Physica E 118 113879
 Google Scholar
Google Scholar
[30] Ma S H, Yuan D Y, Wang Y R, Jiao Z Y 2018 J. Mater. Chem. C 6 8082
 Google Scholar
Google Scholar
[31] Giovannetti G, Khomyakov P A, Brocks G, Karpan V M, Brink J V D, Kelly P J 2008 Phys. Rev. Lett. 101 026803
 Google Scholar
Google Scholar
[32] Zhang J, Yang G, Yuan D, Tian J L, Ma D W 2019 J. Appl. Phys. 125 074501
 Google Scholar
Google Scholar
[33] Pohle R, Tawil A, Davydovskaya P, Fleischer M 2011 Procedia Eng. 25 108
 Google Scholar
Google Scholar
[34] Zhao J, Cui X Y, Huang Q Q, Zeng H 2022 Appl. Surf. Sci. 613 156010
 Google Scholar
Google Scholar
[35] Taylor J, Guo H, Wang J 2001 Phys. Rev. B 63 245407
 Google Scholar
Google Scholar
[36] Brandbyge M, Mozos J L, Ordejón P, Taylor J, Stokbro K 2002 Phys. Rev. B 65 165401
 Google Scholar
Google Scholar
[37] Watanabe K, Taniguchi T, Kanda H 2004 Nat. Mater. 3 404
 Google Scholar
Google Scholar
[38] Huang C S, Murat A, Babar V, Montes E, Schwingenschlögl U 2018 J. Phys. Chem. C 122 14665
 Google Scholar
Google Scholar
[39] Kumar V, Roy D R 2019 Physica E 110 100
 Google Scholar
Google Scholar
[40] Ashori E, Nazari F, Illas F 2014 Int. J. Hydrogen. Energ. 39 6610
 Google Scholar
Google Scholar
[41] Ayesh A I 2022 Superlattice Microst. 162 107098
 Google Scholar
Google Scholar
[42] Xu Y, Jiang S X, Yin W J, Sheng W, Wu L X, Nie G Z, Ao Z M 2020 Appl. Surf. Sci. 501 144199
 Google Scholar
Google Scholar
[43] Xu K, Liao N B, Zheng B R, Zhou H M 2020 Phys. Lett. A 384 126533
 Google Scholar
Google Scholar
[44] Zheng Y P, Li E L, Liu C, Bai K F, Cui Z, Ma D M 2021 J. Phys. Chem. Solids 152 109857
 Google Scholar
Google Scholar
[45] Liu L, Yang Q, Wang Z P, Ye H Y, Chen X P, Fan X J, Zhang G Q 2018 Appl. Surf. Sci. 433 575
 Google Scholar
Google Scholar
[46] Kumar V, Rajput K, Roy D R 2022 Appl. Surf. Sci. 606 154741
 Google Scholar
Google Scholar
[47] 李小林, 袁坤, 何嘉乐, 刘洪峰, 张建波, 周阳 2022 71 017103
 Google Scholar
Google Scholar
Li X L, Yuan K, He J L, Liu H F, Zhang J B, Zhou Y 2022 Acta Phys. Sin. 71 017103
 Google Scholar
Google Scholar
[48] Guo H Y, Zhang W H, Lu N, Zhuo Z W, Zeng X C, Wu X J, Yang J L 2015 J. Phys. Chem. C 119 6912
 Google Scholar
Google Scholar
[49] Wang Y S, Song N H, Song X Y, Zhang T J, Yang D P, Li M 2018 Vacuum 147 18
 Google Scholar
Google Scholar
[50] Yu X F, Li Y C, Cheng J B, Liu Z B, Li Q Z, Li W Z, Yang X, Xiao B 2015 ACS Appl. Mater. Interfaces 7 13707
 Google Scholar
Google Scholar
[51] Sajjad M, Montes E, Singh N, Schwingenschlögl U 2016 Adv. Mater. Interfaces 4 1600911
 Google Scholar
Google Scholar
[52] Meng R S, Cai M, Jiang J K, Liang Q H, Sun X, Yang Q, Tan C J, Chen X P 2016 IEEE Electron. Device Lett. 38 134
 Google Scholar
Google Scholar
[53] Castro Neto A H, Guinea F, Peres N M R, Novoselov K S, Geim A K 2009 Rev. Mod. Phys. 81 109
 Google Scholar
Google Scholar
[54] 崔兴倩, 刘乾, 范志强, 张振华 2020 69 248501
 Google Scholar
Google Scholar
Cui X Q, Liu Q, Fan Z Q, Zhang Z H 2020 Acta Phys. Sin. 69 248501
 Google Scholar
Google Scholar
[55] 吴洪芬, 冯盼君, 张烁, 刘大鹏, 高淼, 闫循旺 2022 71 036801
 Google Scholar
Google Scholar
Wu H F, Feng P J, Zhang S, Liu D P, Gao M, Yan X W 2022 Acta Phys. Sin. 71 036801
 Google Scholar
Google Scholar
[56] Cui Z, Wu H, Yang K Q, Wang X, Lv Y J 2024 Sens. Actuators A Phys. 366 114954
 Google Scholar
Google Scholar
[57] Tit N, Othman W, Shaheen A, Ali M 2018 Sens. Actuators B Chem. 270 167
 Google Scholar
Google Scholar
-
图 1 正交相BN单层原子结构球棍模型的顶视图和侧视图. 绿球和银球分别表示B原子和N原子, 对于气体分子吸附问题, 分子分别放置在3种不同的初始位置(T1, T2, H1), 图中T1和T2分别表示N原子和B原子的顶位, H1表示在六元环中心的空位
Fig. 1. Top view and side views of optimized structure of the pristine BN monolayer. Green and silver balls indicating B and N atoms, respectively. Three different absorption sites are considered, namely, T1, T2, H1, and the gas molecules are initially at these sites. T1 and T2 represent the top sites above the N and B atom, respectively, and H1 represents the hollow site.
图 2 纳米电子器件模型示意图, 它包含左电极、右电极和中心散射区. 左右电极与中心区域无缝连接, 中心区域分子的吸附会对器件的电输运性能产生影响
Fig. 2. Schematic demonstration of the nanodevice consisting of left-, right-electrode and the central region. The left and right electrodes are seamlessly connected with the central region, and the molecule adsorption at the central region will have impacts on the electrical performance of device.
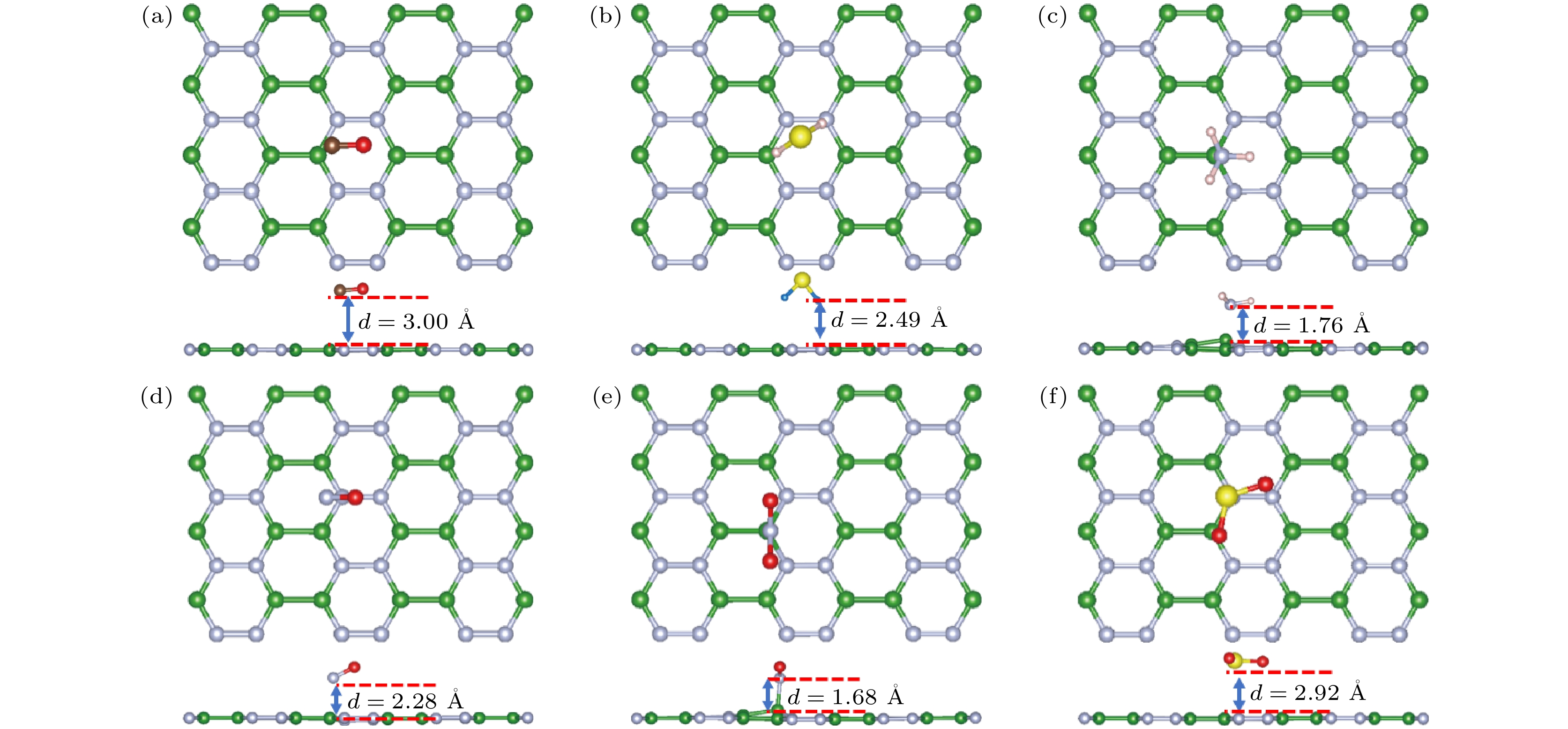
图 4 气体在单层o-BN上稳定吸附构象的俯视图和侧视图 (a) CO; (b) H2S; (c) NH3; (d) NO; (e) NO2; (f) SO2; 绿色、银色、棕色、红色、白色、黄色球分别表示B, N, C, O, H和S原子
Fig. 4. Top and side views of the most stable adsorption sites for (a) CO, (b) H2S, (c) NH3, (d) NO, (e) NO2 and (f) SO2 molecules adsorbed on the monolayered BN. The green, silver, brown, red, white, yellow balls indicate B, N, C, O, H and S atoms, respectively.
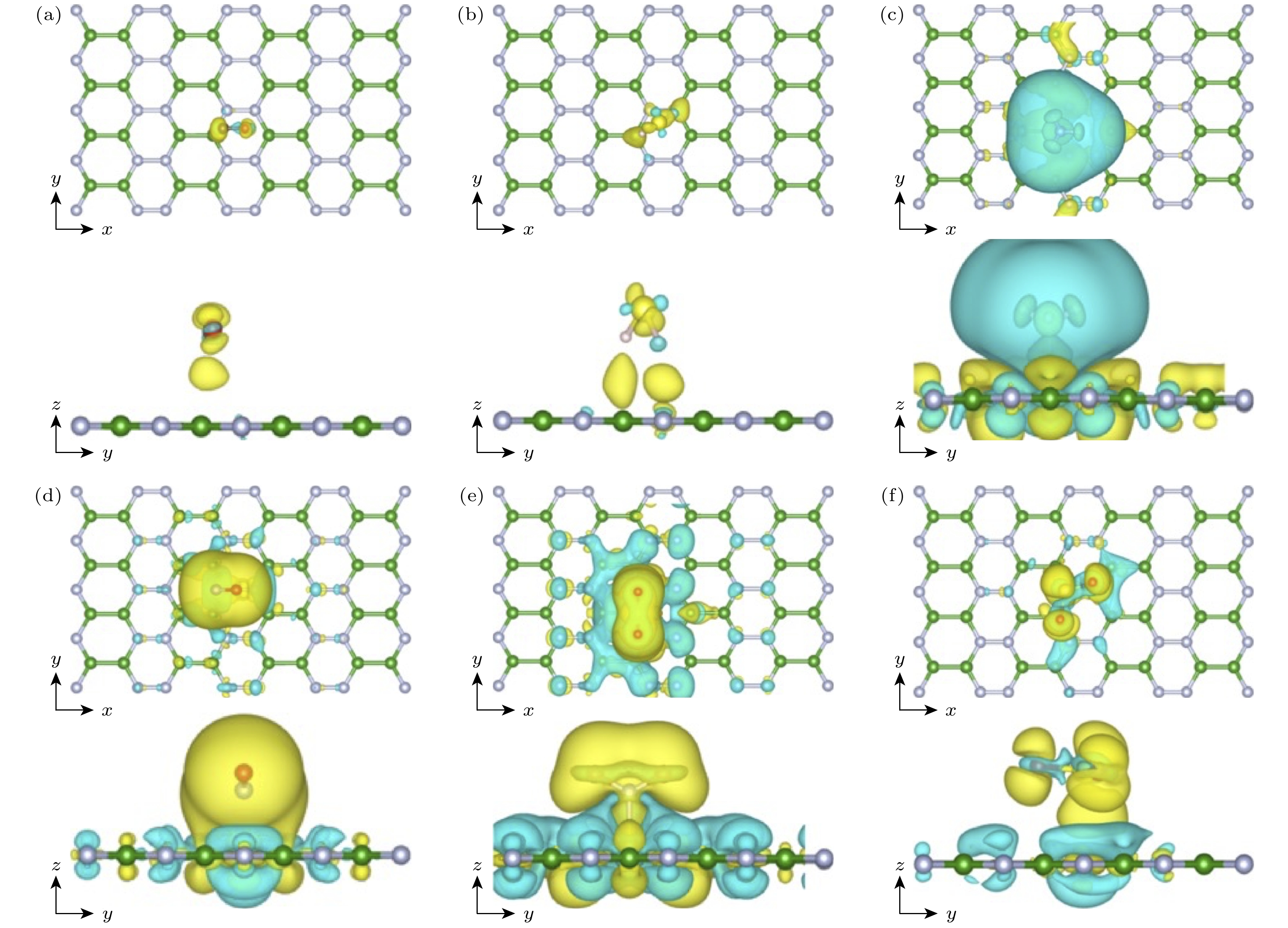
图 6 o-BN单层吸附气体分子的差分电荷密度的俯视图和侧视图 (a) CO; (b) H2S; (c) NH3; (d) NO; (e) NO2; (f) SO2; 其中等值线为0.0005 e·Bohr–3, 黄色和青色分别表示电子聚集和电子耗尽的程度
Fig. 6. Charge density difference contours for (a) CO, (b) H2S, (c) NH3, (d) NO, (e) NO2, and (f) SO2 adsorbed on the o-BN monolayer (0.0003 e·Bohr–3 is set for isosurface value). Yellow and cyan represent electron accumulation and electron depletion, respectively.
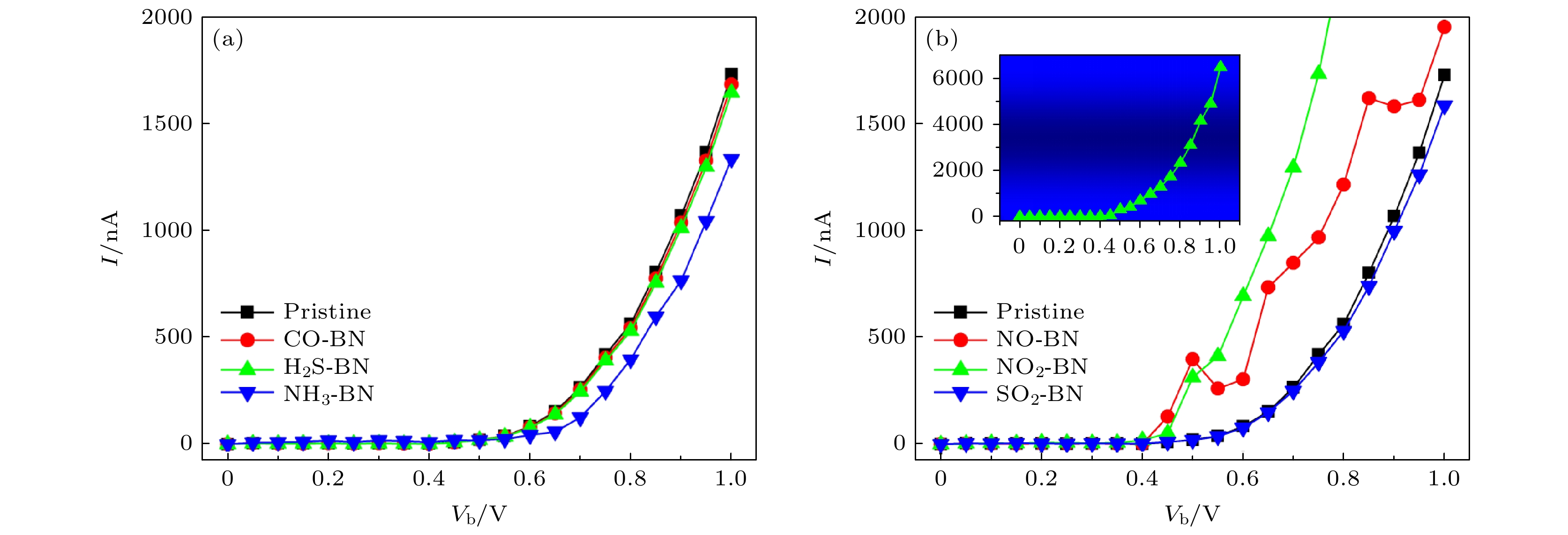
图 10 (a), (b)气体分子吸附前后o-BN单层器件的$ I{\text{-}}{V}_{{\mathrm{b}}} $曲线, 图(b)中插图展示了NO2吸附的o-BN单层器件完整的$ I{\text{-}}{V}_{{\mathrm{b}}} $结果
Fig. 10. Electrical performance of $ I{\text{-}}{V}_{{\mathrm{b}}} $ curves of (a) pristine o-BN monolayer device and (b) gas molecule adsorbed o-BN monolayer devices. The inset in panel (b) exhibits the $ I{\text{-}}{V}_{{\mathrm{b}}} $ curve of the NO2 adsorbed o-BN monolayer device.
表 1 吸附能$ {E}_{{\mathrm{a}}{\mathrm{d}}} $ (eV)、气体与衬底之间的距离d (Å)和电荷转移Q (e), 正值表示气体分子从o-BN衬底获得电子
Table 1. Adsorption energy $ {E}_{{\mathrm{a}}{\mathrm{d}}} $ (eV), distances between gas and the substrate d (Å) and the charge transfer Q (e). The positive value means the gas molecule obtains electron from the BN substrate.
$ {E}_{{\mathrm{a}}{\mathrm{d}}} $/eV d/Å Q/e CO –0.151 2.999 0.032 H2S –0.186 2.488 0.014 NH3 –0.222 1.762 –0.102 NO –0.258 2.284 0.133 NO2 –0.893 1.679 0.780 SO2 –0.380 2.922 0.165 表 2 气体在300—600 K之间吸附后的恢复时间τ(s)
Table 2. Recovery time τ(s) after gas adsorption ranging from 300 to 600 K.
Gas
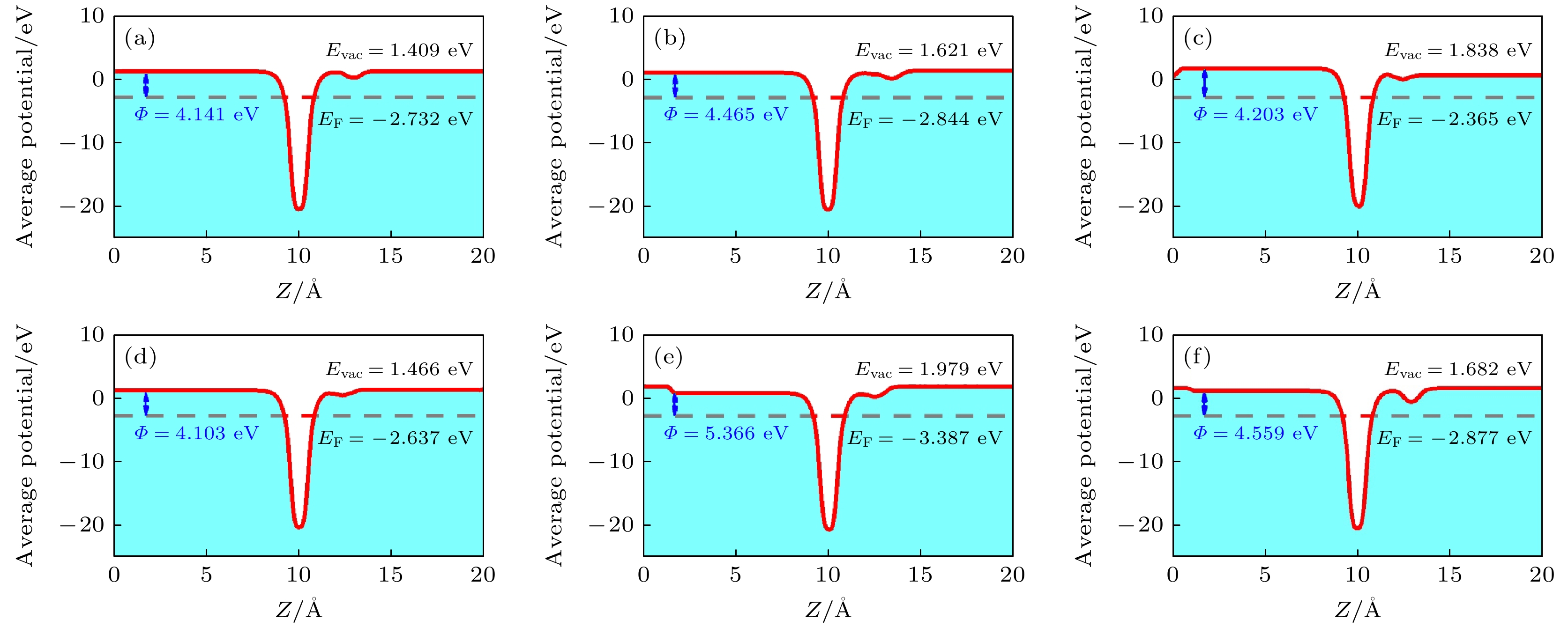
moleculeTemperature/K 300 400 500 600 CO 3.44×10–11 7.99×10–12 3.33×10–12 1.85×10–12 H2S 1.33×10–10 2.20×10–11 7.49×10–12 3.65×10–12 NH3 5.36×10–10 6.26×10–11 1.73×10–11 7.32×10–12 NO 2.16×10–9 1.78×10–10 3.98×10–11 1.47×10–11 NO2 100.13 0.02 1.00×10–4 3.16×10–6 SO2 2.42×10–7 6.13×10–9 6.76×10–10 1.55×10–10 表 3 气体分子吸附在o-BN单层后的功函数$ \varPhi $(eV)、功函数差$ {{\Delta }}\varPhi $(eV)和灵敏度S(%)
Table 3. Work function $ \varPhi~{\mathrm{(eV)}}$, work function difference $ {{\Delta }}\varPhi $ (eV) and sensitivity S (%) of the gas molecules adsorbed on the o-BN monolayer.
Adsorbed gas $ \varPhi $/eV $ {{\Delta }}\varPhi $/eV S/% CO 4.141 0.032 0.78 H2S 4.465 0.356 8.66 NH3 4.203 0.094 2.29 NO 4.103 –0.006 0.15 NO2 5.366 1.257 30.59 SO2 4.599 0.450 10.95 -
[1] Radisavljevic B, Radenovic A, Brivio J, Giacometti V, Kis A 2011 Nat. Nanotechnol. 6 147
 Google Scholar
Google Scholar
[2] Tombros N, Jozsa C, Popinciuc M, Jonkman H T, Wees B 2007 Nature 448 571
 Google Scholar
Google Scholar
[3] Allen M, Tung V, Kaner R 2010 Chem. Rev. 110 132
 Google Scholar
Google Scholar
[4] Wang G Y, Yu J B, Zheng K, Huang Y X, Li X D, Chen X P, Tao L Q 2020 IEEE Electron. Device Lett. 41 1404
 Google Scholar
Google Scholar
[5] Ma S X, Li D J, Rao X J, Xia X F, Su Y, Lu Y F 2020 Adsorption 26 619
 Google Scholar
Google Scholar
[6] Lin L Y, Liu T M, Zhang Y, Sun R, Zeng W, Wang Z C 2016 Ceram. Int. 42 971
 Google Scholar
Google Scholar
[7] Feng C, Qin H B, Yang D G, Zhang G Q 2019 Materials 12 676
 Google Scholar
Google Scholar
[8] Ren J, Kong W, Ni J 2019 Nanoscale Res. Lett. 14 1
 Google Scholar
Google Scholar
[9] Sun X, Yang Q, Meng R S, Tan C J, Liang Q H, Jiang J K, Ye H Y, Chen X P 2017 Appl. Surf. Sci. 404 291
 Google Scholar
Google Scholar
[10] Shahriar R, Hassan O, Alam M K 2022 RSC Adv. 12 16732
 Google Scholar
Google Scholar
[11] 徐强, 段康, 谢浩, 张秦蓉, 梁本权, 彭祯凯, 李卫 2021 70 157101
 Google Scholar
Google Scholar
Xu Q, Duan K, Xie H, Zhang Q R, Liang B Q, Peng Z K, Li W 2021 Acta Phys. Sin. 70 157101
 Google Scholar
Google Scholar
[12] Zhang D Z, Pan W J, Tang M C, Wang D Y, Yu S J, Mi Q, Pan Q N, Hu Y Q 2023 Nano Res. 16 11959
 Google Scholar
Google Scholar
[13] Demirci S, Rad S E, Kazak S, Nezir S, Jahangirov S 2020 Phys. Rev. B 101 125408
 Google Scholar
Google Scholar
[14] Zhao J, Zeng H, Yao G 2021 Phys. Chem. Chem. Phys. 23 3771
 Google Scholar
Google Scholar
[15] Rahimi R, Solimannejad M 2021 Energy Fuels 35 6858
 Google Scholar
Google Scholar
[16] Khossossi N, Luo W, Haman Z, Singh D, Essaoudi I, Ainane A, Ahuja R 2022 Nano Energy 96 107066
 Google Scholar
Google Scholar
[17] Ma D W, Lu Z S, Ju W W, et al. 2012 J. Phys.: Condens. Mat. 24 145501
 Google Scholar
Google Scholar
[18] Zhou Y G, Dong J X, Wang Z G, Xiao H Y, Gao F, Zu X T 2010 Phys. Chem. Chem. Phys. 12 7588
 Google Scholar
Google Scholar
[19] Zhou Y G, Zu X T, Yang P, et al. 2010 J. Phys.: Condens. Mat. 22 465303
 Google Scholar
Google Scholar
[20] Li J, Hu M L, Yu Z Z, Zhong J X, Sun L Z 2012 Chem. Phys. Lett. 532 40
 Google Scholar
Google Scholar
[21] Xu R H, Kong F, Wan J, Guo J Y 2023 Mat. Sci. Semicon. Proc. 161 107446
 Google Scholar
Google Scholar
[22] Paghi A, Mariani S, Barillaro G 2023 Small 19 2206100
 Google Scholar
Google Scholar
[23] Yang S, Jiang C, Wei S H 2017 Appl. Phys. Rev. 4 021304
 Google Scholar
Google Scholar
[24] Kresse G, Hafner J 1993 Phys. Rev. B 47 558
 Google Scholar
Google Scholar
[25] Kresse G, Furthmuller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[26] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[27] Grimme S 2006 J. Comput. Chem. 27 1787
 Google Scholar
Google Scholar
[28] Monkhorst H J, Pack J D 1976 Phys. Rev. B 13 5188
 Google Scholar
Google Scholar
[29] Zhang J N, Ma L, Zhang M, Zhang J M 2020 Physica E 118 113879
 Google Scholar
Google Scholar
[30] Ma S H, Yuan D Y, Wang Y R, Jiao Z Y 2018 J. Mater. Chem. C 6 8082
 Google Scholar
Google Scholar
[31] Giovannetti G, Khomyakov P A, Brocks G, Karpan V M, Brink J V D, Kelly P J 2008 Phys. Rev. Lett. 101 026803
 Google Scholar
Google Scholar
[32] Zhang J, Yang G, Yuan D, Tian J L, Ma D W 2019 J. Appl. Phys. 125 074501
 Google Scholar
Google Scholar
[33] Pohle R, Tawil A, Davydovskaya P, Fleischer M 2011 Procedia Eng. 25 108
 Google Scholar
Google Scholar
[34] Zhao J, Cui X Y, Huang Q Q, Zeng H 2022 Appl. Surf. Sci. 613 156010
 Google Scholar
Google Scholar
[35] Taylor J, Guo H, Wang J 2001 Phys. Rev. B 63 245407
 Google Scholar
Google Scholar
[36] Brandbyge M, Mozos J L, Ordejón P, Taylor J, Stokbro K 2002 Phys. Rev. B 65 165401
 Google Scholar
Google Scholar
[37] Watanabe K, Taniguchi T, Kanda H 2004 Nat. Mater. 3 404
 Google Scholar
Google Scholar
[38] Huang C S, Murat A, Babar V, Montes E, Schwingenschlögl U 2018 J. Phys. Chem. C 122 14665
 Google Scholar
Google Scholar
[39] Kumar V, Roy D R 2019 Physica E 110 100
 Google Scholar
Google Scholar
[40] Ashori E, Nazari F, Illas F 2014 Int. J. Hydrogen. Energ. 39 6610
 Google Scholar
Google Scholar
[41] Ayesh A I 2022 Superlattice Microst. 162 107098
 Google Scholar
Google Scholar
[42] Xu Y, Jiang S X, Yin W J, Sheng W, Wu L X, Nie G Z, Ao Z M 2020 Appl. Surf. Sci. 501 144199
 Google Scholar
Google Scholar
[43] Xu K, Liao N B, Zheng B R, Zhou H M 2020 Phys. Lett. A 384 126533
 Google Scholar
Google Scholar
[44] Zheng Y P, Li E L, Liu C, Bai K F, Cui Z, Ma D M 2021 J. Phys. Chem. Solids 152 109857
 Google Scholar
Google Scholar
[45] Liu L, Yang Q, Wang Z P, Ye H Y, Chen X P, Fan X J, Zhang G Q 2018 Appl. Surf. Sci. 433 575
 Google Scholar
Google Scholar
[46] Kumar V, Rajput K, Roy D R 2022 Appl. Surf. Sci. 606 154741
 Google Scholar
Google Scholar
[47] 李小林, 袁坤, 何嘉乐, 刘洪峰, 张建波, 周阳 2022 71 017103
 Google Scholar
Google Scholar
Li X L, Yuan K, He J L, Liu H F, Zhang J B, Zhou Y 2022 Acta Phys. Sin. 71 017103
 Google Scholar
Google Scholar
[48] Guo H Y, Zhang W H, Lu N, Zhuo Z W, Zeng X C, Wu X J, Yang J L 2015 J. Phys. Chem. C 119 6912
 Google Scholar
Google Scholar
[49] Wang Y S, Song N H, Song X Y, Zhang T J, Yang D P, Li M 2018 Vacuum 147 18
 Google Scholar
Google Scholar
[50] Yu X F, Li Y C, Cheng J B, Liu Z B, Li Q Z, Li W Z, Yang X, Xiao B 2015 ACS Appl. Mater. Interfaces 7 13707
 Google Scholar
Google Scholar
[51] Sajjad M, Montes E, Singh N, Schwingenschlögl U 2016 Adv. Mater. Interfaces 4 1600911
 Google Scholar
Google Scholar
[52] Meng R S, Cai M, Jiang J K, Liang Q H, Sun X, Yang Q, Tan C J, Chen X P 2016 IEEE Electron. Device Lett. 38 134
 Google Scholar
Google Scholar
[53] Castro Neto A H, Guinea F, Peres N M R, Novoselov K S, Geim A K 2009 Rev. Mod. Phys. 81 109
 Google Scholar
Google Scholar
[54] 崔兴倩, 刘乾, 范志强, 张振华 2020 69 248501
 Google Scholar
Google Scholar
Cui X Q, Liu Q, Fan Z Q, Zhang Z H 2020 Acta Phys. Sin. 69 248501
 Google Scholar
Google Scholar
[55] 吴洪芬, 冯盼君, 张烁, 刘大鹏, 高淼, 闫循旺 2022 71 036801
 Google Scholar
Google Scholar
Wu H F, Feng P J, Zhang S, Liu D P, Gao M, Yan X W 2022 Acta Phys. Sin. 71 036801
 Google Scholar
Google Scholar
[56] Cui Z, Wu H, Yang K Q, Wang X, Lv Y J 2024 Sens. Actuators A Phys. 366 114954
 Google Scholar
Google Scholar
[57] Tit N, Othman W, Shaheen A, Ali M 2018 Sens. Actuators B Chem. 270 167
 Google Scholar
Google Scholar
计量
- 文章访问数: 2939
- PDF下载量: 125
- 被引次数: 0














 下载:
下载: