-
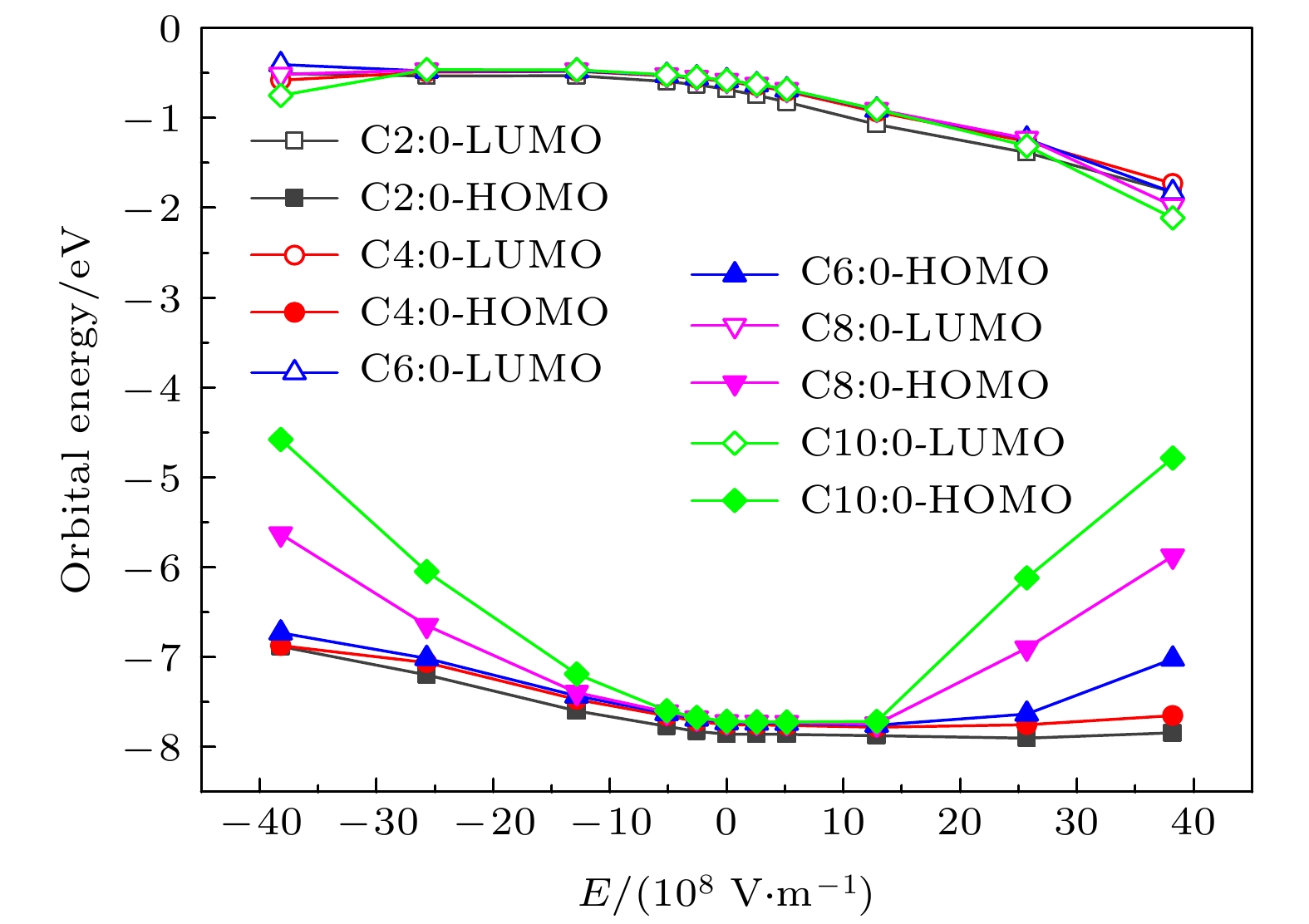
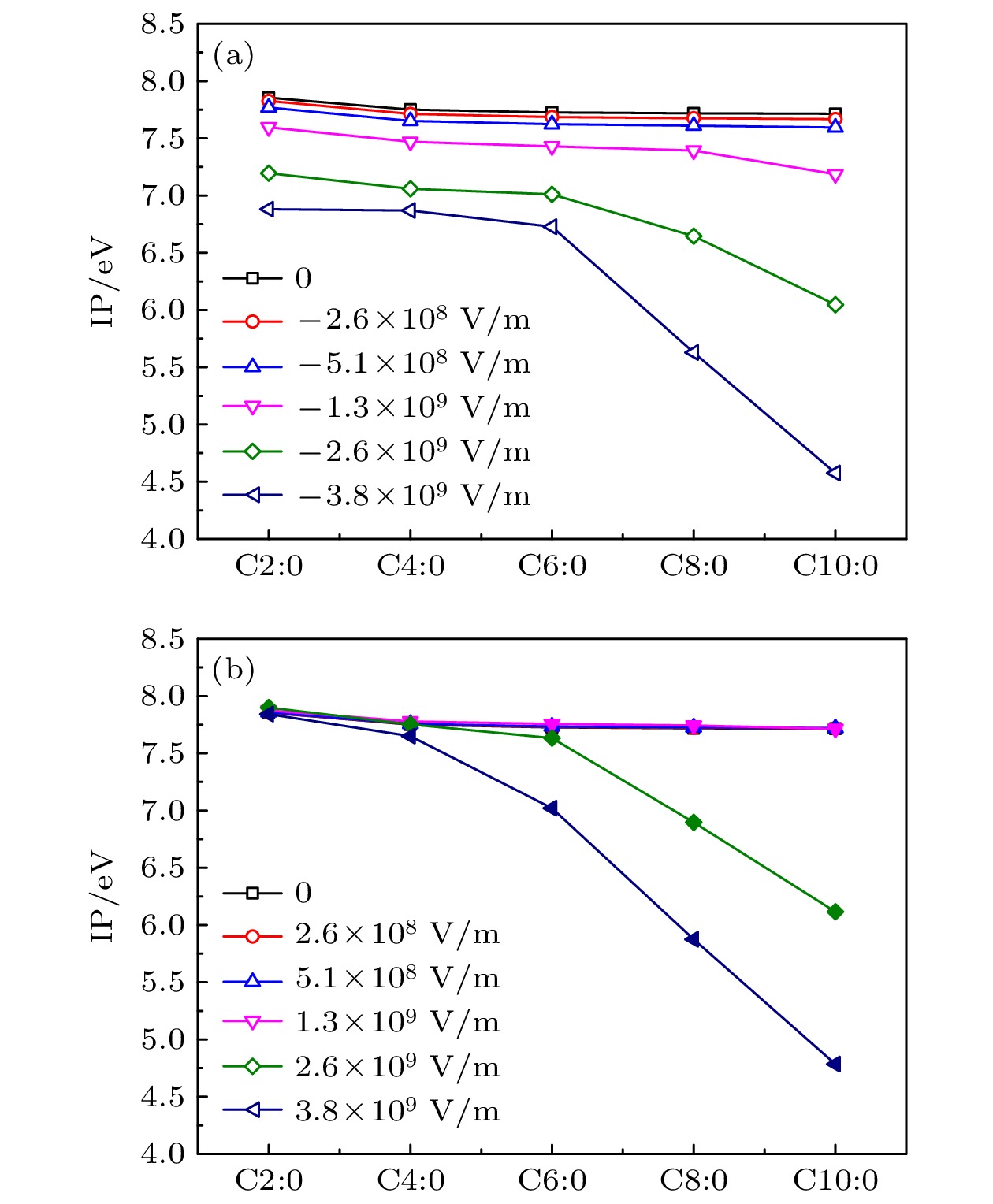
短-中链饱和甘油三酯是一种黏度低、可降解的液体绝缘材料, 在变压器内绝缘领域具有潜在的应用价值. 本文通过设置与绝缘油中放电相当的电场强度等级, 使用密度泛函与含时密度泛函方法研究了短-中链饱和甘油三酯分子在电场影响下的分子特性变化规律. 研究结果表明, 电场下分子结构发生明显改变, 分子键长与电场存在明显的依赖关系; 相同电场下, 随着碳链长度的增大, 分子的偶极矩增大, 分子极性增强; 在109 V/m量级的电场强度下, 三辛酸甘油酯与三癸酸甘油酯的最高占据态分子轨道能量明显增大, 电离势急剧减小; 分子间的激发特性差异较小, 都随着电场强度的增大呈现出减小趋势, 且相同电场下分子激发能的减小幅度远小于电离势的减小幅度. 研究结果有助于提高人们对酯类绝缘介质中放电机理的认识, 并为天然酯绝缘油的性能改进提供一定的理论支撑.Short-medium chain saturated triglyceride is a low viscosity and degradable liquid dielectric material, which has potential applications in the field of transformer internal insulation. In this paper, the molecular properties of short-medium chain saturated triglycerides under the action of electric field are studied by using density functional theory and time-dependent density functional theory. The results show that the molecular bond length is obviously dependent on the electric field intensity, which is consistent with the shift of the infrared wave number peak. Under the same electric field, with the increase of the length of carbon chain, the dipole moment and polarity of the molecule increase correspondingly. When the electric field intensity is 109 V/m, the energy of the highest occupied molecular orbital of tricaprylin and tricaprin increases obviously, and the ionization potential decreases sharply. The difference in excitation characteristic between molecules is small, and the decrease of excitation energy is much smaller than that of ionization potential under the same electric field. The results are helpful in improving the understanding of discharge mechanism in ester dielectric, and provide the theoretical support for the performance improvement of natural ester insulating oil.
-
Keywords:
- triglyceride /
- density functional theory /
- ionization potential /
- excited state
[1] Dong L, Zhong X, He J, Zhang L, Huang X 2016 Clin. Nutr. 35 399
 Google Scholar
Google Scholar
[2] Heerdt B G, Houston M A, Anthony G M, Augenlicht L H 1999 Cancer Res. 59 1584
[3] Fofana I 2013 IEEE Electr. Insul. Mag. 29 13
 Google Scholar
Google Scholar
[4] Wedin P 2014 IEEE Electr. Insul. Mag. 30 20
 Google Scholar
Google Scholar
[5] Thakur S, Sarathi R, Danikas M G 2019 Electr. Eng. 101 1007
 Google Scholar
Google Scholar
[6] Dombek G, Gielniak J 2018 IEEE Trans. Dielectr. Electr. Insul. 5 1846
 Google Scholar
Google Scholar
[7] Trnka P, Hornak J, Prosr P, Michal O, Wang F 2020 IEEE Access 8 61989
 Google Scholar
Google Scholar
[8] Rozga P 2016 IET Sci. Meas. Technol. 10 665
 Google Scholar
Google Scholar
[9] Rodríguez M, Galán M 1995 Chem. Eng. J. 60 117
 Google Scholar
Google Scholar
[10] Tobazcon R 1994 IEEE Trans. Dielectr. Electr. Insul. 1 1132
 Google Scholar
Google Scholar
[11] Beroual A, Zahn M, Badent A, Kist K, Torshin Y 1998 IEEE Electr. Insul. Mag. 14 6
 Google Scholar
Google Scholar
[12] Rozga P 2015 IEEE Trans. Dielectr. Electr. Insul. 22 2754
 Google Scholar
Google Scholar
[13] Li J, Wang Y C, Wang F P, Liang S N, Lin X, Chen X P, Zhou J H 2017 Phys. Lett. A 381 3732
 Google Scholar
Google Scholar
[14] Wang Y C, Wang F P, Li J, Liang S N, Zhou J H 2018 Energies 11 523
 Google Scholar
Google Scholar
[15] Smalo H S, Hestad Ø, Ingebrigtsen S, Åstrand P O 2011 J. Appl. Phys. 109 073306
 Google Scholar
Google Scholar
[16] Wang Y C, Wang F P, Li J, Huang Z Y, Liang S N, Zhou J H 2017 Energies 10 510
 Google Scholar
Google Scholar
[17] 黄多辉, 王藩侯, 程晓洪, 万明杰 蒋刚 2011 60 123101
 Google Scholar
Google Scholar
Huang D H, Wang P H, Cheng X H, Wang M J, Jiang G 2011 Acta Phys. Sin. 60 123101
 Google Scholar
Google Scholar
[18] Xu G L, Liu X F, Xie H X, Zhang X Z, Liu Y F 2010 Chin. Phys. B 19 113201
 Google Scholar
Google Scholar
[19] 曹欣伟, 任杨, 刘慧, 李姝丽 2014 63 043101
 Google Scholar
Google Scholar
Cao X W, Ren Y, Liu H, Li S L 2014 Acta Phys. Sin. 63 043101
 Google Scholar
Google Scholar
[20] Xu G L, Xie H X, Wei Y, Zhang X Z Liu Y F 2012 Chin. Phys. B 21 153
 Google Scholar
Google Scholar
[21] Grozema F C, Telesca R, Jonkman H T, Siebbeles L, Snijders J G 2001 J. Chem. Phys. 115 10014
 Google Scholar
Google Scholar
[22] 杜建宾, 武德起, 唐延林, 隆正文 2015 64 073101
 Google Scholar
Google Scholar
Du J B, Wu D Q, Tang Y L, Long Z W 2015 Acta Phys. Sin. 64 073101
 Google Scholar
Google Scholar
[23] 袁伟 2013 硕士学位论文 (新乡: 河南师范大学)
Yuan W 2013 M. S. Thesis (Xinxiang: Henan Normal University) (in Chinese)
[24] NIST Computational Chemistry Comparison and Benchmark Data Basehttp://cccbdb.nist.gov/vibscalejust.asp [2020-8-1]
[25] NIST Standard Reference Database 69: Chemistry WebBook https://webbook.nist.gov/chemistry/ [2018-10-1]
[26] 凌智钢, 唐延林, 李涛, 李玉鹏, 魏晓楠 2013 62 223102
 Google Scholar
Google Scholar
Ling Z G, Tang Y L, Li T, Li Y P, Wei X N 2013 Acta Phys. Sin. 62 223102
 Google Scholar
Google Scholar
[27] 李晓虎 2006 博士学位论文 (重庆: 重庆大学)
Li X H 2006 Ph. D. Dissertation (Chongqing: Chongqing University) (in Chinese)
[28] 李世雄, 吴永刚, 令狐荣锋, 孙光宇, 张正平 秦水介 2015 64 043101
 Google Scholar
Google Scholar
Li S X, Wu Y G, Linghu R F, Sun G Y, Zhang Z P, Qing S J 2015 Acta Phys. Sin. 64 043101
 Google Scholar
Google Scholar
[29] Li J, Liu X Y, Zhu Z H, Sheng Y 2012 Chin. Phys. B 21 033101
 Google Scholar
Google Scholar
[30] Lu T, Chen F W 2012 J. Comput. Chem. 33 580
 Google Scholar
Google Scholar
[31] Smalo H S, Astrand P O Ingebrigtsen S 2010 IEEE Trans. Dielectr. Electr. Insul. 17 733
 Google Scholar
Google Scholar
[32] Jadidian J, Zahn M, Lavesson N, Widlund O, Borg K 2012 IEEE Trans. Plasma Sci. 40 909
 Google Scholar
Google Scholar
-
表 1 不同计算方法的比较
Table 1. Comparison of different calculation methods
方法 甘油
三酯波数 v/ cm–1 C—O—O C=O C—H C2:0 1235.8 1816.3 2938.4 C4:0 1185.9 1809.5 2938.3 HF/631+
G*/0.90C6:0 1162 1809.2 2926.4 C8:0 1179.5 1809.1 2926.2 C10:0 1160.2 1808.9 2908.9 C2:0 1173.9 1787.1 2602.1 C4:0 1131.2 1778.3 2993.7 B3LYP/631G*/0.96 C6:0 1126.8 1778.2 2983.1 C8:0 1125.2 1778.2 2981.7 C10:0 1089.3 1778.2 2907.7 C2:0 1181.6 1756.8 2956.7 C4:0 1123.3 1750.7 2987.5 B3LYP/631+G*/0.96 C6:0 1119.1 1750.5 2975.6 C8:0 1117.6 1750.4 2940.5 C10:0 1116.5 1750.4 2943.9 C2:0 1164.1 1751.3 2936.3 C4:0 1107.6 1745.4 2966.1 B3LYP/6311++G**/0.96 C6:0 1104.2 1745.1 2959.5 C8:0 1103 1745 2895.9 C10:0 1102.3 1745 2929.9 C2:0 1208.0 1738.6 2950.0 实验值 C4:0 1159.0 1736.5 2962.5 C6:0 1159.8 1744.4 2950.4 C8:0 1155.5 1747.5 2958.3 C10:0 − − − 表 2 电场下C6∶0分子的键长
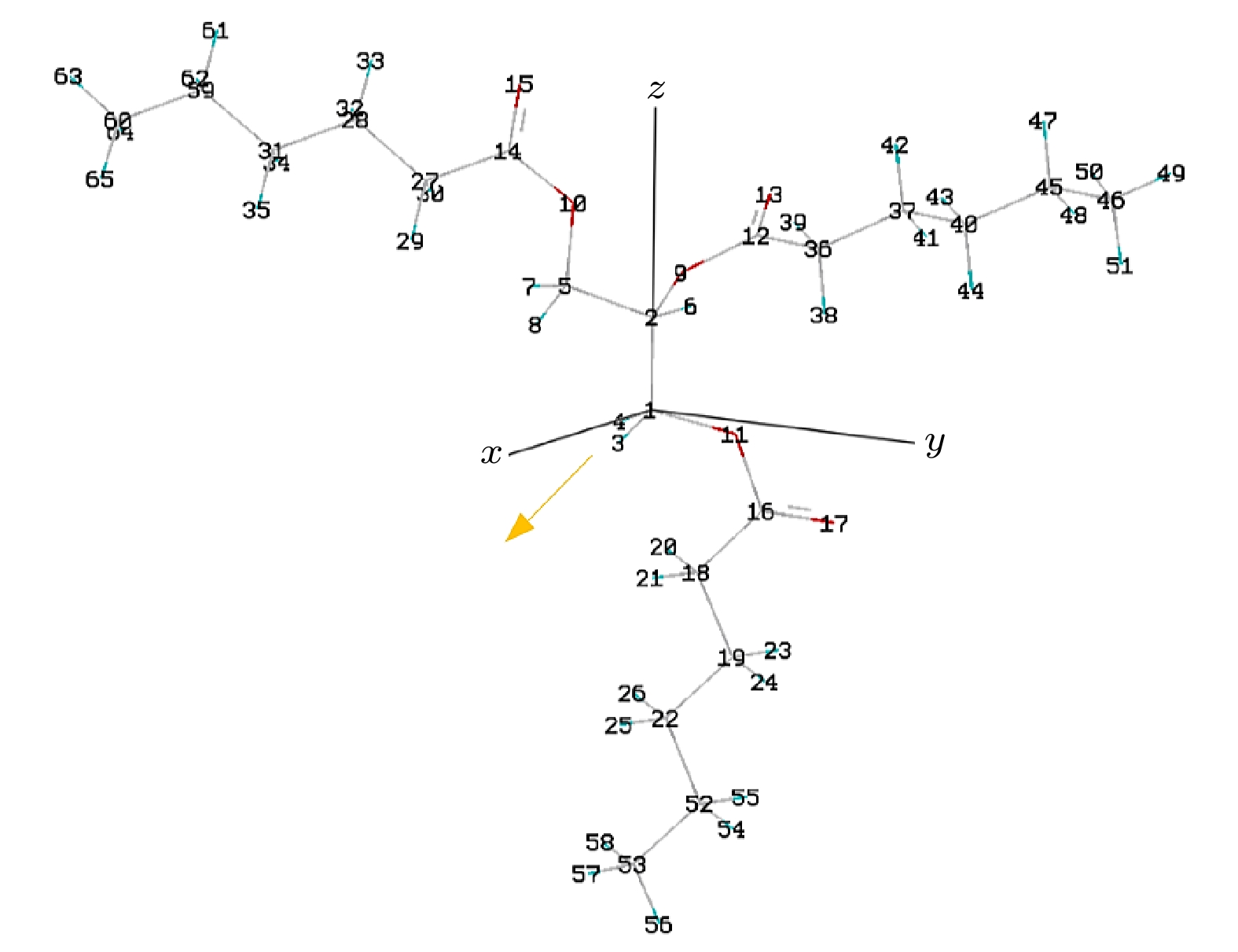
Table 2. Bond length of C6∶0 molecule under electric field.
E/(108 V·m–1) 2C—9O/Å 12C—9O/Å 12C=13O/Å 5C—10O/Å 14C—10O/Å 14C = 15O/Å 1C—11O/Å 16C=17O/Å –38 1.435 1.389 1.205 1.434 1.379 1.209 1.438 1.214 –25 1.434 1.388 1.207 1.433 1.379 1.207 1.432 1.211 –13 1.434 1.384 1.207 1.431 1.376 1.207 1.430 1.209 –5.1 1.434 1.381 1.207 1.431 1.373 1.206 1.429 1.208 –2.6 1.434 1.380 1.207 1.432 1.372 1.208 1.428 1.208 0 1.434 1.380 1.207 1.433 1.371 1.207 1.428 1.207 2.6 1.434 1.379 1.207 1.433 1.370 1.208 1.428 1.207 5.1 1.434 1.378 1.207 1.434 1.369 1.208 1.428 1.207 13 1.435 1.375 1.208 1.436 1.366 1.210 1.428 1.206 25 1.434 1.373 1.209 1.439 1.360 1.213 1.428 1.205 38 1.434 1.370 1.211 1.443 1.355 1.216 1.425 1.203 表 3 电场下C6∶0分子的前线轨道组分
Table 3. The frontier orbital composition of C6∶0 molecule under electric field.
E/(108 V·m–1) Composition of the HOMO and LUMO (>3%) –38 HOMO 5C∶3.73, 12C∶4.49, 37C∶3.35, 40C∶11.88, 43H∶4.08, 45C∶18.02, 46C∶13.24, 47H∶4.2, 48H∶10.08, 50H∶6.99 LUMO 1C∶18.58, 2C∶18.88, 5C∶31.84, 18C∶6.56, 19C∶10.27, 22C∶6.87 –5.1 HOMO 2C∶3.07, 12C∶3.36, 13O∶34.35, 36C∶25.08, 37C∶5.25, 40C∶4.97 LUMO 1C∶7.12, 2C∶7.25, 14C∶5.28, 18C∶25.35, 19C∶33.40, 27C∶6.48, 28C3.29, 31C∶3.17 0 HOMO 2C∶5.86, 5C∶5.03, 11O∶7.80, 17O∶31.35, 18C∶17.39, 19C∶5.15, 22C∶5.58 LUMO 1C∶5.77, 2C∶6.75, 14C∶4.68, 18C∶26.92, 19C∶32.22, 27C∶7.36, 28C∶4.81, 31C∶3.87 5.1 HOMO 2C∶5.62, 5C∶4.92, 11O∶8.19, 12C∶3.31, 16C∶3.17, 17O∶32.67, 18C∶17.61, 19C∶5.21, 22C∶5.88 LUMO 1C∶8.10, 2C∶9.33, 14C∶3.39, 18C∶7.20, 19C∶29.10, 27C∶6.93, 28C∶4.89, 31C∶4.08 38 HOMO 1C∶15.52, 2C∶31.37, 18C∶5.46, 19C∶7.80, 52C∶8.47, 53C∶6.22, 54H∶4.66, 55H∶5.03, 57H∶3.53, 58H∶3.32 LUMO 27C∶9.74, 28C∶26.00, 31C∶33.47, 59C∶26.02 表 4 电场下分子的激发态
Table 4. The molecular excited state under electric field.
分子 E /(108 V·m–1) m = 1 m = 2 m = 3 m = 4 m = 5 m = 6 m = 7 m = 8 m = 9 0 Eex /eV 5.6219 5.6422 5.6657 6.6449 6.6683 6.7503 6.7754 6.8532 6.8874 λ /nm 220.54 219.74 218.83 186.59 185.93 183.67 182.99 180.91 180.02 f 0.0009 0.0009 0.0004 0.0056 0.0015 0.0177 0.0096 0.0019 0.0032 5.1 Eex /eV 5.6149 5.6418 5.6619 6.6358 6.6848 6.7356 6.7626 6.8506 6.8776 C2∶0 λ /nm 220.81 219.76 218.98 186.84 185.47 184.07 183.34 180.98 180.27 f 0.0009 0.0009 0.0005 0.0071 0.0009 0.0169 0.0098 0.0041 0.0022 26 Eex /eV 5.5859 5.6366 5.643 6.3958 6.4734 6.5507 6.7110 6.8013 6.8396 λ /nm 221.96 219.96 219.71 193.85 191.53 189.27 184.75 182.30 181.27 f 0.0008 0.0009 0.0004 0.0040 0.0024 0.0070 0.0005 0.0227 0.0023 0 Eex /eV 5.6609 5.6751 5.7014 6.6073 6.6248 6.7205 6.7478 6.8553 6.8990 λ /nm 219.02 218.47 217.46 187.65 187.15 184.49 183.74 180.86 179.71 f 0.0004 0.0005 0.0002 0.0017 0.0115 0.0113 0.0004 0.0013 0.0055 5.1 Eex /eV 5.6551 5.6747 5.6991 6.6070 6.6271 6.7076 6.7391 6.8510 6.8983 C6∶0 λ /nm 219.24 218.49 217.55 187.66 187.09 184.84 183.98 180.97 179.73 f 0.0004 0.0005 0.0002 0.0127 0.0006 0.0110 0.0002 0.0051 0.0017 26 Eex /eV 5.6314 5.6697 5.6808 6.3806 6.4593 6.5178 6.7284 6.7854 6.8576 λ /nm 220.17 218.68 218.25 194.31 191.95 190.22 184.27 182.72 180.80 f 0.0004 0.0004 0.0001 0.0062 0.0011 0.0037 0.0004 0.0123 0.0011 0 Eex /eV 5.6594 5.6735 5.7005 6.6005 6.6186 6.7133 6.7432 6.8516 6.8955 λ /nm 219.08 218.53 217.5 187.84 187.33 184.68 183.87 180.96 179.80 f 0.0004 0.0005 0.0002 0.0017 0.0124 0.0117 0.0004 0.0014 0.0056 5.1 Eex /eV 5.6538 5.6734 5.6984 6.5991 6.6206 6.7002 6.7336 6.8443 6.8952 C10∶0 λ /nm 219.29 218.54 217.58 187.88 187.27 185.05 184.13 181.15 179.81 f 0.0004 0.0005 0.0002 0.0136 0.0005 0.0115 0.0002 0.0052 0.0019 26 Eex /eV 5.6303 5.6676 5.6812 6.3752 6.4561 6.5099 6.7246 6.7745 6.8546 λ /nm 220.21 218.76 218.24 194.48 192.04 190.45 184.37 183.01 180.88 f 0.0004 0.0004 0.0001 0.0067 0.0010 0.0040 0.0005 0.0132 0.0010 -
[1] Dong L, Zhong X, He J, Zhang L, Huang X 2016 Clin. Nutr. 35 399
 Google Scholar
Google Scholar
[2] Heerdt B G, Houston M A, Anthony G M, Augenlicht L H 1999 Cancer Res. 59 1584
[3] Fofana I 2013 IEEE Electr. Insul. Mag. 29 13
 Google Scholar
Google Scholar
[4] Wedin P 2014 IEEE Electr. Insul. Mag. 30 20
 Google Scholar
Google Scholar
[5] Thakur S, Sarathi R, Danikas M G 2019 Electr. Eng. 101 1007
 Google Scholar
Google Scholar
[6] Dombek G, Gielniak J 2018 IEEE Trans. Dielectr. Electr. Insul. 5 1846
 Google Scholar
Google Scholar
[7] Trnka P, Hornak J, Prosr P, Michal O, Wang F 2020 IEEE Access 8 61989
 Google Scholar
Google Scholar
[8] Rozga P 2016 IET Sci. Meas. Technol. 10 665
 Google Scholar
Google Scholar
[9] Rodríguez M, Galán M 1995 Chem. Eng. J. 60 117
 Google Scholar
Google Scholar
[10] Tobazcon R 1994 IEEE Trans. Dielectr. Electr. Insul. 1 1132
 Google Scholar
Google Scholar
[11] Beroual A, Zahn M, Badent A, Kist K, Torshin Y 1998 IEEE Electr. Insul. Mag. 14 6
 Google Scholar
Google Scholar
[12] Rozga P 2015 IEEE Trans. Dielectr. Electr. Insul. 22 2754
 Google Scholar
Google Scholar
[13] Li J, Wang Y C, Wang F P, Liang S N, Lin X, Chen X P, Zhou J H 2017 Phys. Lett. A 381 3732
 Google Scholar
Google Scholar
[14] Wang Y C, Wang F P, Li J, Liang S N, Zhou J H 2018 Energies 11 523
 Google Scholar
Google Scholar
[15] Smalo H S, Hestad Ø, Ingebrigtsen S, Åstrand P O 2011 J. Appl. Phys. 109 073306
 Google Scholar
Google Scholar
[16] Wang Y C, Wang F P, Li J, Huang Z Y, Liang S N, Zhou J H 2017 Energies 10 510
 Google Scholar
Google Scholar
[17] 黄多辉, 王藩侯, 程晓洪, 万明杰 蒋刚 2011 60 123101
 Google Scholar
Google Scholar
Huang D H, Wang P H, Cheng X H, Wang M J, Jiang G 2011 Acta Phys. Sin. 60 123101
 Google Scholar
Google Scholar
[18] Xu G L, Liu X F, Xie H X, Zhang X Z, Liu Y F 2010 Chin. Phys. B 19 113201
 Google Scholar
Google Scholar
[19] 曹欣伟, 任杨, 刘慧, 李姝丽 2014 63 043101
 Google Scholar
Google Scholar
Cao X W, Ren Y, Liu H, Li S L 2014 Acta Phys. Sin. 63 043101
 Google Scholar
Google Scholar
[20] Xu G L, Xie H X, Wei Y, Zhang X Z Liu Y F 2012 Chin. Phys. B 21 153
 Google Scholar
Google Scholar
[21] Grozema F C, Telesca R, Jonkman H T, Siebbeles L, Snijders J G 2001 J. Chem. Phys. 115 10014
 Google Scholar
Google Scholar
[22] 杜建宾, 武德起, 唐延林, 隆正文 2015 64 073101
 Google Scholar
Google Scholar
Du J B, Wu D Q, Tang Y L, Long Z W 2015 Acta Phys. Sin. 64 073101
 Google Scholar
Google Scholar
[23] 袁伟 2013 硕士学位论文 (新乡: 河南师范大学)
Yuan W 2013 M. S. Thesis (Xinxiang: Henan Normal University) (in Chinese)
[24] NIST Computational Chemistry Comparison and Benchmark Data Basehttp://cccbdb.nist.gov/vibscalejust.asp [2020-8-1]
[25] NIST Standard Reference Database 69: Chemistry WebBook https://webbook.nist.gov/chemistry/ [2018-10-1]
[26] 凌智钢, 唐延林, 李涛, 李玉鹏, 魏晓楠 2013 62 223102
 Google Scholar
Google Scholar
Ling Z G, Tang Y L, Li T, Li Y P, Wei X N 2013 Acta Phys. Sin. 62 223102
 Google Scholar
Google Scholar
[27] 李晓虎 2006 博士学位论文 (重庆: 重庆大学)
Li X H 2006 Ph. D. Dissertation (Chongqing: Chongqing University) (in Chinese)
[28] 李世雄, 吴永刚, 令狐荣锋, 孙光宇, 张正平 秦水介 2015 64 043101
 Google Scholar
Google Scholar
Li S X, Wu Y G, Linghu R F, Sun G Y, Zhang Z P, Qing S J 2015 Acta Phys. Sin. 64 043101
 Google Scholar
Google Scholar
[29] Li J, Liu X Y, Zhu Z H, Sheng Y 2012 Chin. Phys. B 21 033101
 Google Scholar
Google Scholar
[30] Lu T, Chen F W 2012 J. Comput. Chem. 33 580
 Google Scholar
Google Scholar
[31] Smalo H S, Astrand P O Ingebrigtsen S 2010 IEEE Trans. Dielectr. Electr. Insul. 17 733
 Google Scholar
Google Scholar
[32] Jadidian J, Zahn M, Lavesson N, Widlund O, Borg K 2012 IEEE Trans. Plasma Sci. 40 909
 Google Scholar
Google Scholar
计量
- 文章访问数: 5865
- PDF下载量: 95
- 被引次数: 0













 下载:
下载: