-
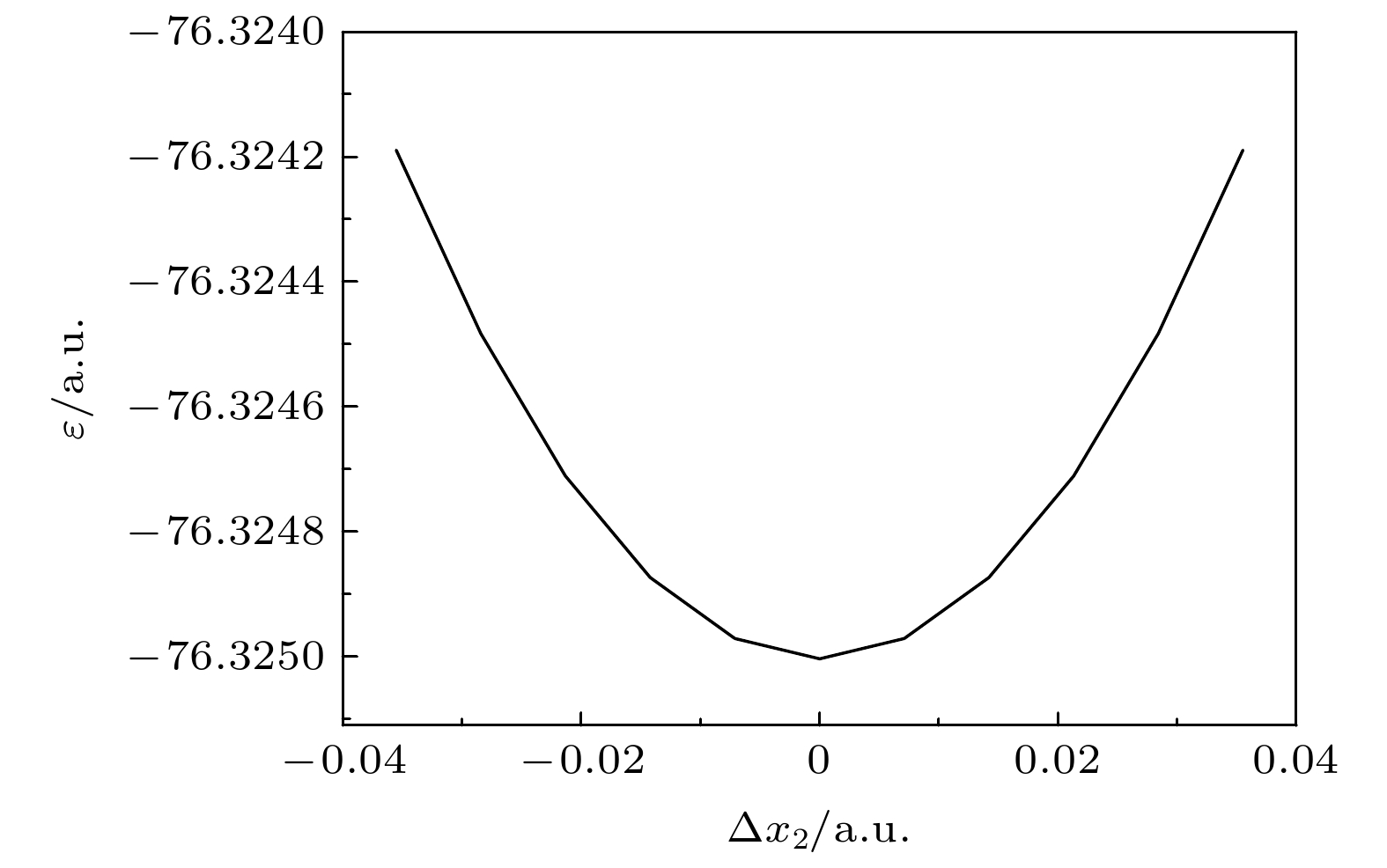
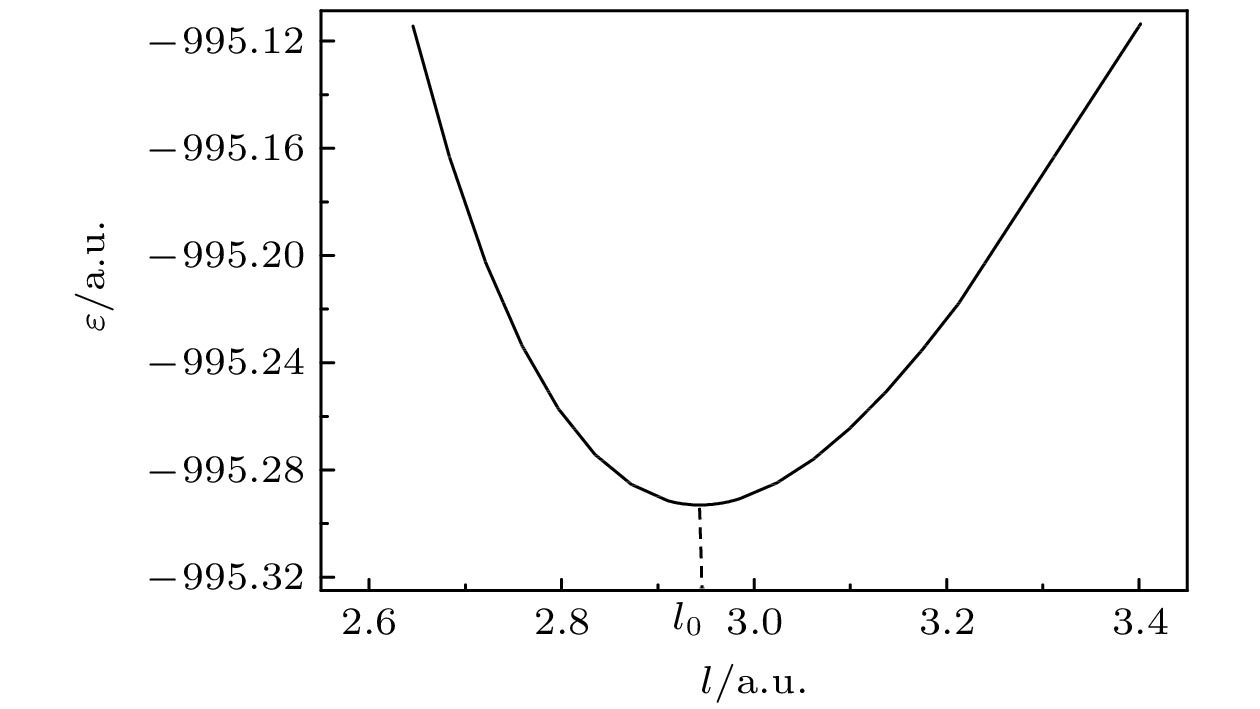
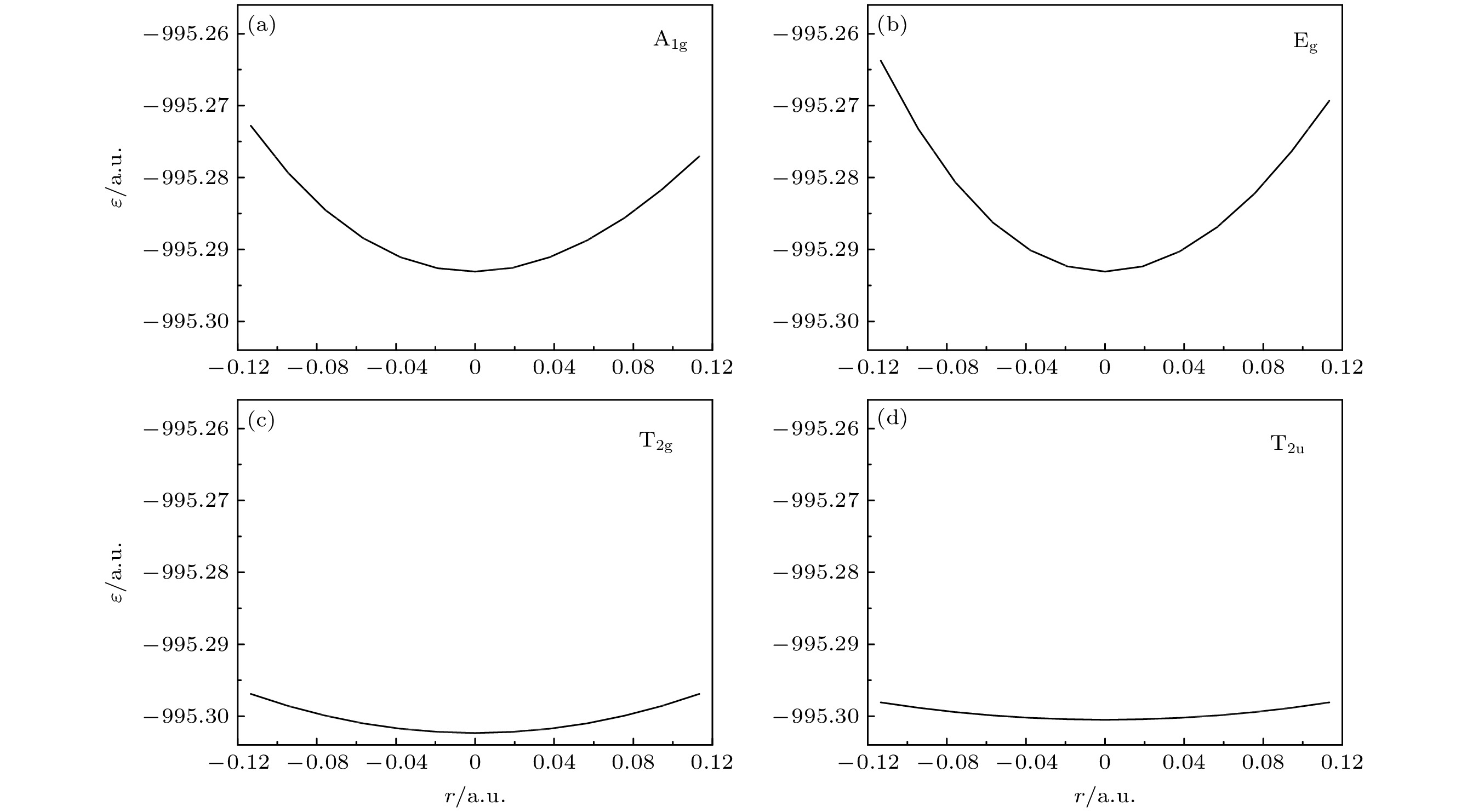
Quantum calculation of molecular vibrational frequency is important in investigating infrared spectrum and Raman spectrum. In this work, a low computational cost method of calculating the quantum chemistry of vibrational frequencies for large molecules is proposed. Usually, the calculation of vibrational frequency of a molecule containing N atoms needs to deal with the Hessian matrix, which consists of second derivatives of the 3N-dimensional potential hypersurface, and then solve secular equations of the matrix to obtain normal vibration modes and the corresponding frequencies. Larger N implies higher computational cost. Therefore, for a limited computational hardware condition, higher-level computations for large N atomic molecule’s vibrational frequencies cannot be implemented in practice. Here we solve this problem by calculating the vibrational frequency for only one vibrational mode each time instead of calculating the Hessian matrix to obtain all vibrational frequencies. When only one vibrational mode is taken into consideration, the molecular potential hypersurface can be transformed into one-dimensional curve. Hence, we can calculate the curve with high-level computational method, then deduce the expression of one-dimensional curve by using harmonic oscillating approximation and obtain the vibrational frequency by using the expression to fit the curve. It should be noted that this method is applied to vibrational modes whose vibrational coordinates can be completely determined by equilibrium geometry and the molecular symmetry and be independent of the molecular force constants. It requires that there exists no other vibrational mode with the same symmetry but with different frequencies. The lower computational cost for a one-dimensional potential curve than that for 3N-dimensional potential hypersurface’s second derivatives permits us to use higher-level method and larger basis set for a given computational hardware condition to achieve more accurate results. In this paper we take the calculation of B2 vibrational frequency of water molecule for example to illustrate the feasibility of this method. Furthermore, we use this method to deal with the SF6 molecule. It has 7 atoms and 70 electrons, hence there exists a large amount of electronic correlation energy to be calculated. The MRCI is an effective method to calculate the correlation energy. But by now no MRCI result of SF6 vibrational frequencies has been reported. So here we use MRCI/6-311G* to calculate the potential curves of A1g, Eg, T2g and T2u vibrational modes separately, deduce their expressions, then use the expressions to fit the curves, and finally obtain the vibrational frequencies. The results are then compared with those obtained by other theoretical methods including HF, MP2, CISD, CCSD(T) and B3LYP methods through using the same 6-311G* basis set. It is shown that the relative error to experimental result of the MRCI method is the least in the results from all these methods.
-
Keywords:
- quantum chemical calculation /
- molecular vibration /
- molecular symmetry /
- potential function
[1] 徐光宪, 黎乐民, 王德民 2009 量子化学(中)(第二版) (北京: 科学出版社) 第342, 343页
Xu G X, Li L M, Wang D M 2009 Quantum Chemistry (Vol. 2) (2nd Ed.) (Beijing: Science Press) pp342, 343 (in Chinese)
[2] 李晨曦, 郭迎春, 王兵兵 2017 66 103101
 Google Scholar
Google Scholar
Li C X, Guo Y C, Wang B B 2017 Acta Phys. Sin. 66 103101
 Google Scholar
Google Scholar
[3] Johnson R D Computational Chemistry Comparison and Benchmark Data Base, https://cccbdb.nist.gov/vibs1x.asp [2021-11-10]
[4] 周朕蕊, 韩冬, 赵明月, 张国强 2020 电工技术学报 35 4998
 Google Scholar
Google Scholar
Zhou L R, Han D, Zhao M Y, Zhang G Q 2020 Trans. Chin. Electrotech. Soc. 35 4998
 Google Scholar
Google Scholar
[5] Okubo H, Beroual A 2011 IEEE Electr. Insul. Mag. 27 34
 Google Scholar
Google Scholar
[6] Zhang X, Yu L, Gui Y, Hu W 2016 Appl. Surf. Sci. 367 259
 Google Scholar
Google Scholar
[7] 武瑞琪, 郭迎春, 王兵兵 2019 68 080201
 Google Scholar
Google Scholar
Wu R Q, Guo Y C, Wang B B 2019 Acta Phys. Sin. 68 080201
 Google Scholar
Google Scholar
[8] Ferré A, Boguslavskiy A E, Dagan M, Blanchet V, Bruner B D, Burgy F, Camper A, Descamps D, Fabre B, Fedorov N, Gaudin J, Geoffroy G, Mikosch J, Patchkovskii S, Petit S, Ruchon T, Soifer H, Staedter D, Wilkinson I, Stolow A, Dudovich N, Mairesse Y 2015 Nat. Commun. 6 5952
 Google Scholar
Google Scholar
[9] Wagner N L, Wuest A, Christov I P, Popmintchev T, Zhou X, Murnane M M, Kapteyn H C 2006 Proc. Natl. Acad. Sci. U. S. A. 103 13279
 Google Scholar
Google Scholar
[10] Jose J, Lucchese R R 2015 Chem. Phys. 447 64
 Google Scholar
Google Scholar
[11] Nguyen N T, Lucchese R R, Lin C D, Le A T 2016 Phys. Rev. A 93 063419
 Google Scholar
Google Scholar
[12] McDowell, R S, Krohn B J, Flicker H, Vasquez M C 1986 Spectrochim. Acta, Part A 42 351
 Google Scholar
Google Scholar
[13] Chrysos M, Rachet F, Kremer D 2014 J. Chem. Phys. 140 124308
 Google Scholar
Google Scholar
[14] Faye M, Boudon V, Loëte M 2016 J. Mol. Spectrosc. 325 35
 Google Scholar
Google Scholar
[15] Chapados C, Birnbaum G 1988 J. Mol. Spectrosc. 132 323
 Google Scholar
Google Scholar
[16] Kremer D, Rachet F, Chrysos M 2013 J. Chem. Phys. 138 174308
 Google Scholar
Google Scholar
[17] Eisfeld W 2011 J. Chem. Phys. 134 054303
 Google Scholar
Google Scholar
[18] Bin T, Longfei Z, Fangyuan H, Zongchang L, Qinqin L, Chenyao L, Liping Z, Jieming Z 2018 AIP Adv. 8 015016
 Google Scholar
Google Scholar
[19] Watanabe N, Hirayama T, Takahashi M 2019 Phys. Rev. A 99 062708
 Google Scholar
Google Scholar
[20] Werner H J, Knowles P J, Knizia G, Manby F R, Schütz M 2012 Wiley Interdiscip. Rev. Comput. Mol. Sci. 2 242
 Google Scholar
Google Scholar
[21] Werner H J, Knowles P J, Manby F, Black J A, Doll K, Hebelmann A, Kats D, Köhn A, Korona T, Kreplin D A, Ma Q, Miller T F, Mitrushchenkov A, Peterson K A, Polyak I, Rauhut G, Sibaev M R 2020 J. Chem. Phys. 152 144107
 Google Scholar
Google Scholar
[22] 巴音贺希格 1996 大学物理 8 12
 Google Scholar
Google Scholar
Bayanheshig 1996 College Physics 8 12
 Google Scholar
Google Scholar
-
表 1 SF6的A1g, Eg, T2g和T2u振动模式
Table 1. A1g, Eg, T2g and T2u vibrational modes of SF6 molecule.
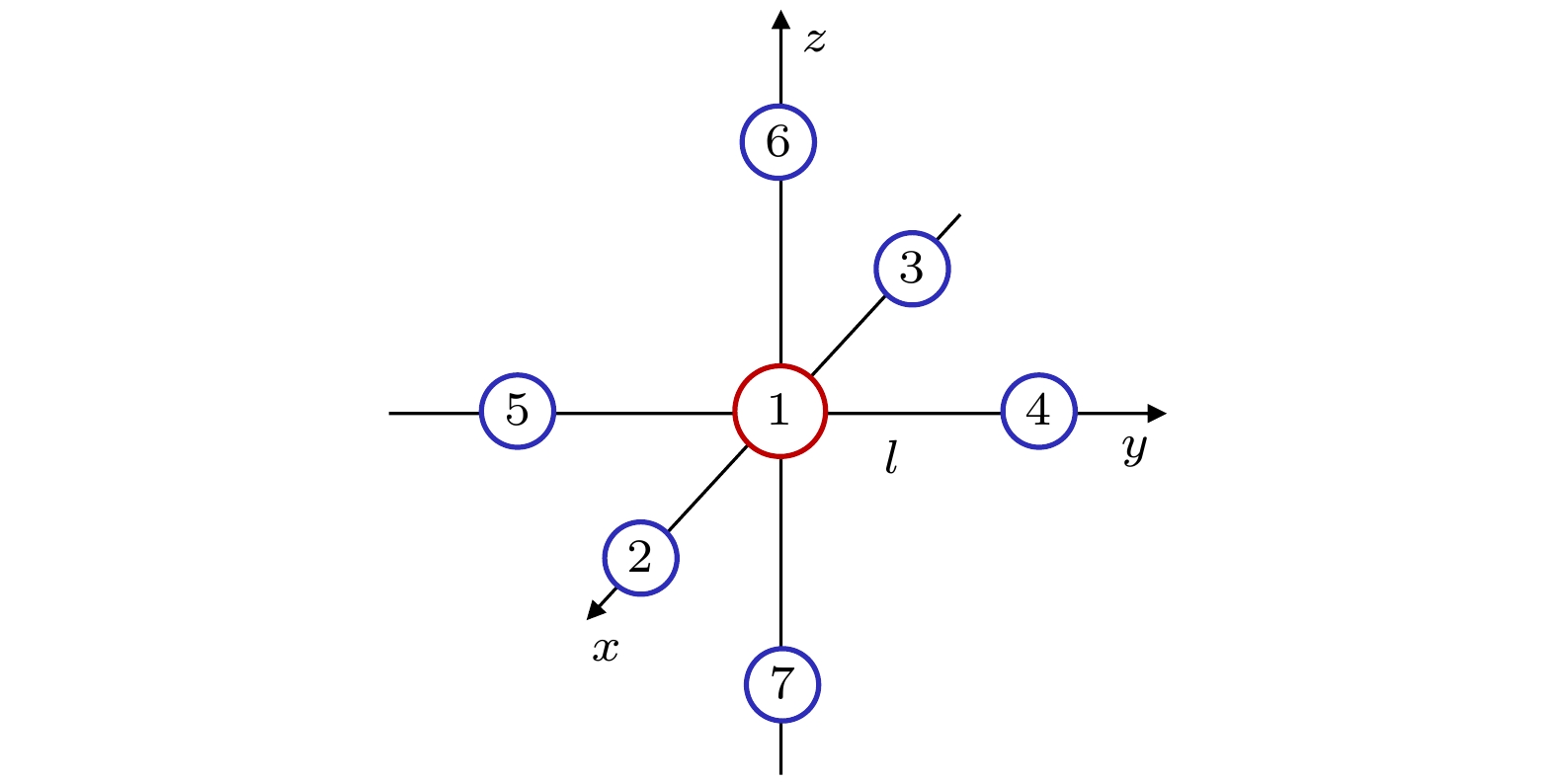
位移坐标 $ {\mathrm{A}}_{1\mathrm{g}} $ $ {\mathrm{E}}_{\mathrm{g}} $ $ {\mathrm{E}}_{\mathrm{g}} $ $ {\mathrm{T}}_{2\mathrm{g}} $ $ {\mathrm{T}}_{2\mathrm{g}} $ $ {\mathrm{T}}_{2\mathrm{g}} $ $ {\mathrm{T}}_{2\mathrm{u}} $ $ {\mathrm{T}}_{2\mathrm{u}} $ $ {\mathrm{T}}_{2\mathrm{u}} $ $ {\Delta x}_{1}, \Delta {y}_{1}, \Delta {z}_{1} $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, 0 $ $ {\Delta x}_{2}, \Delta {y}_{2}, \Delta {z}_{2} $ $ r, \mathrm{0, 0} $ ${r}, \mathrm{0, 0}$ $ -r, \mathrm{0, 0} $ $ 0, r, 0 $ $ \mathrm{0, 0}, r $ $ \mathrm{0, 0}, 0 $ $ 0, r, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, r $ $ {\Delta x}_{3}, {\Delta y}_{3}, \Delta {z}_{3} $ $ -r, \mathrm{0, 0} $ $ -r, \mathrm{0, 0} $ $ \mathrm{r}, \mathrm{0, 0} $ $ 0, -r, 0 $ $ \mathrm{0, 0}, -r $ $ \mathrm{0, 0}, 0 $ $ 0, r, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, r $ $ {\Delta x}_{4}, \Delta {y}_{4}, \Delta {z}_{4} $ $ 0, r, 0 $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 2}r, 0 $ $ r, \mathrm{0, 0} $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, r $ $ \mathrm{0, 0}, 0 $ $ r, \mathrm{0, 0} $ $ \mathrm{0, 0}, -r $ $ \Delta {x}_{5}, \Delta {y}_{5}, \Delta {z}_{5} $ $0,-r, 0$ $ \mathrm{0, 0}, 0 $ $ 0, -2 r, 0 $ $ -r, \mathrm{0, 0} $ $ \mathrm{0, 0}, 0 $ $ \mathrm{0, 0}, -r $ $ \mathrm{0, 0}, 0 $ $ r, \mathrm{0, 0} $ $ \mathrm{0, 0}, -r $ $ {\Delta x}_{6}, \Delta {y}_{6}, \Delta {z}_{6} $ $ \mathrm{0, 0}, r $ $ \mathrm{0, 0}, -r $ $ \mathrm{0, 0}, -r $ $ \mathrm{0, 0}, 0 $ $ r, \mathrm{0, 0} $ $ 0, r, 0 $ $ 0, -r, 0 $ $ -r, \mathrm{0, 0} $ $ \mathrm{0, 0}, 0 $ $ {\Delta x}_{7}, \Delta {y}_{7}, \Delta {z}_{7} $ $ \mathrm{0, 0}, -r $ $ \mathrm{0, 0}, r $ $ \mathrm{0, 0}, r $ $ \mathrm{0, 0}, 0 $ $ -r, \mathrm{0, 0} $ $ 0, -r, 0 $ $ 0, -r, 0 $ $ -r, \mathrm{0, 0} $ $ \mathrm{0, 0}, 0 $ 表 2 SF6的A1g, Eg, T2g和T2u振动模式的振动频率ω1, ω2, ω3和ω4
Table 2. Vibrational frequencies ω1, ω2, ω3 and ω4 of the vibrational modes A1g, Eg, T2g and T2u of SF6
MRCI/6-311G* HF
/6-311G*MP2/6-311G* CISD/6-311G* CCSD(T)/6-311G* B3LYP/6-311g* Expt[12] $ {\omega }_{1}/\mathrm{c}{\mathrm{m}}^{-1} $ 809 824 726 806 719 706 787 $ {\omega }_{2}/\mathrm{c}{\mathrm{m}}^{-1} $ 650 697 625 684 627 611 655 $ {\omega }_{3}/\mathrm{c}{\mathrm{m}}^{-1} $ 542 588 502 541 500 467 524 $ {\omega }_{4}/\mathrm{c}{\mathrm{m}}^{-1} $ 362 377 335 364 334 311 355 l0/(10–10 m) 1.558 1.547 1.586 1.557 1.586 1.593 1.565 -
[1] 徐光宪, 黎乐民, 王德民 2009 量子化学(中)(第二版) (北京: 科学出版社) 第342, 343页
Xu G X, Li L M, Wang D M 2009 Quantum Chemistry (Vol. 2) (2nd Ed.) (Beijing: Science Press) pp342, 343 (in Chinese)
[2] 李晨曦, 郭迎春, 王兵兵 2017 66 103101
 Google Scholar
Google Scholar
Li C X, Guo Y C, Wang B B 2017 Acta Phys. Sin. 66 103101
 Google Scholar
Google Scholar
[3] Johnson R D Computational Chemistry Comparison and Benchmark Data Base, https://cccbdb.nist.gov/vibs1x.asp [2021-11-10]
[4] 周朕蕊, 韩冬, 赵明月, 张国强 2020 电工技术学报 35 4998
 Google Scholar
Google Scholar
Zhou L R, Han D, Zhao M Y, Zhang G Q 2020 Trans. Chin. Electrotech. Soc. 35 4998
 Google Scholar
Google Scholar
[5] Okubo H, Beroual A 2011 IEEE Electr. Insul. Mag. 27 34
 Google Scholar
Google Scholar
[6] Zhang X, Yu L, Gui Y, Hu W 2016 Appl. Surf. Sci. 367 259
 Google Scholar
Google Scholar
[7] 武瑞琪, 郭迎春, 王兵兵 2019 68 080201
 Google Scholar
Google Scholar
Wu R Q, Guo Y C, Wang B B 2019 Acta Phys. Sin. 68 080201
 Google Scholar
Google Scholar
[8] Ferré A, Boguslavskiy A E, Dagan M, Blanchet V, Bruner B D, Burgy F, Camper A, Descamps D, Fabre B, Fedorov N, Gaudin J, Geoffroy G, Mikosch J, Patchkovskii S, Petit S, Ruchon T, Soifer H, Staedter D, Wilkinson I, Stolow A, Dudovich N, Mairesse Y 2015 Nat. Commun. 6 5952
 Google Scholar
Google Scholar
[9] Wagner N L, Wuest A, Christov I P, Popmintchev T, Zhou X, Murnane M M, Kapteyn H C 2006 Proc. Natl. Acad. Sci. U. S. A. 103 13279
 Google Scholar
Google Scholar
[10] Jose J, Lucchese R R 2015 Chem. Phys. 447 64
 Google Scholar
Google Scholar
[11] Nguyen N T, Lucchese R R, Lin C D, Le A T 2016 Phys. Rev. A 93 063419
 Google Scholar
Google Scholar
[12] McDowell, R S, Krohn B J, Flicker H, Vasquez M C 1986 Spectrochim. Acta, Part A 42 351
 Google Scholar
Google Scholar
[13] Chrysos M, Rachet F, Kremer D 2014 J. Chem. Phys. 140 124308
 Google Scholar
Google Scholar
[14] Faye M, Boudon V, Loëte M 2016 J. Mol. Spectrosc. 325 35
 Google Scholar
Google Scholar
[15] Chapados C, Birnbaum G 1988 J. Mol. Spectrosc. 132 323
 Google Scholar
Google Scholar
[16] Kremer D, Rachet F, Chrysos M 2013 J. Chem. Phys. 138 174308
 Google Scholar
Google Scholar
[17] Eisfeld W 2011 J. Chem. Phys. 134 054303
 Google Scholar
Google Scholar
[18] Bin T, Longfei Z, Fangyuan H, Zongchang L, Qinqin L, Chenyao L, Liping Z, Jieming Z 2018 AIP Adv. 8 015016
 Google Scholar
Google Scholar
[19] Watanabe N, Hirayama T, Takahashi M 2019 Phys. Rev. A 99 062708
 Google Scholar
Google Scholar
[20] Werner H J, Knowles P J, Knizia G, Manby F R, Schütz M 2012 Wiley Interdiscip. Rev. Comput. Mol. Sci. 2 242
 Google Scholar
Google Scholar
[21] Werner H J, Knowles P J, Manby F, Black J A, Doll K, Hebelmann A, Kats D, Köhn A, Korona T, Kreplin D A, Ma Q, Miller T F, Mitrushchenkov A, Peterson K A, Polyak I, Rauhut G, Sibaev M R 2020 J. Chem. Phys. 152 144107
 Google Scholar
Google Scholar
[22] 巴音贺希格 1996 大学物理 8 12
 Google Scholar
Google Scholar
Bayanheshig 1996 College Physics 8 12
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 12164
- PDF Downloads: 242
- Cited By: 0















 DownLoad:
DownLoad: