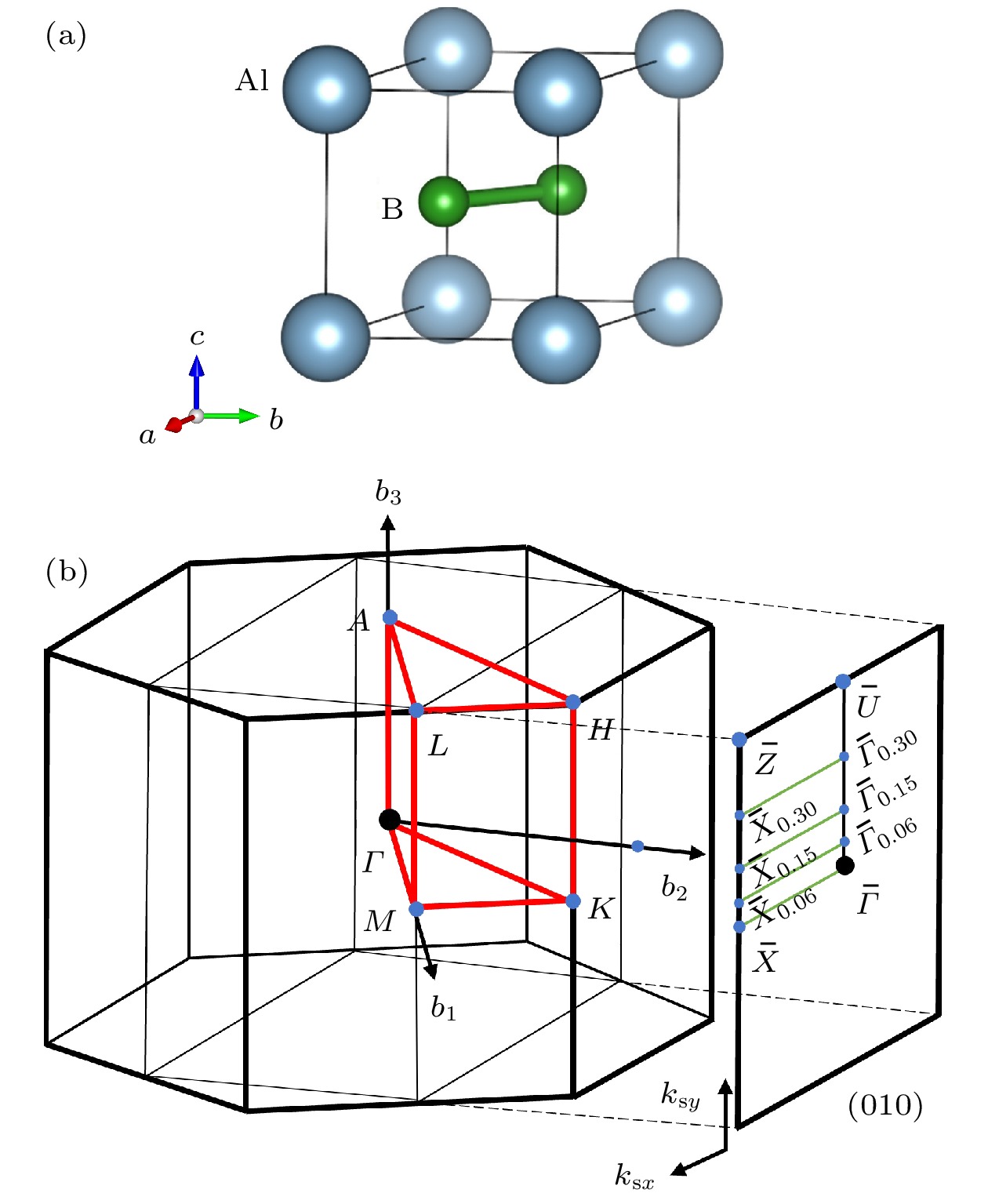
-
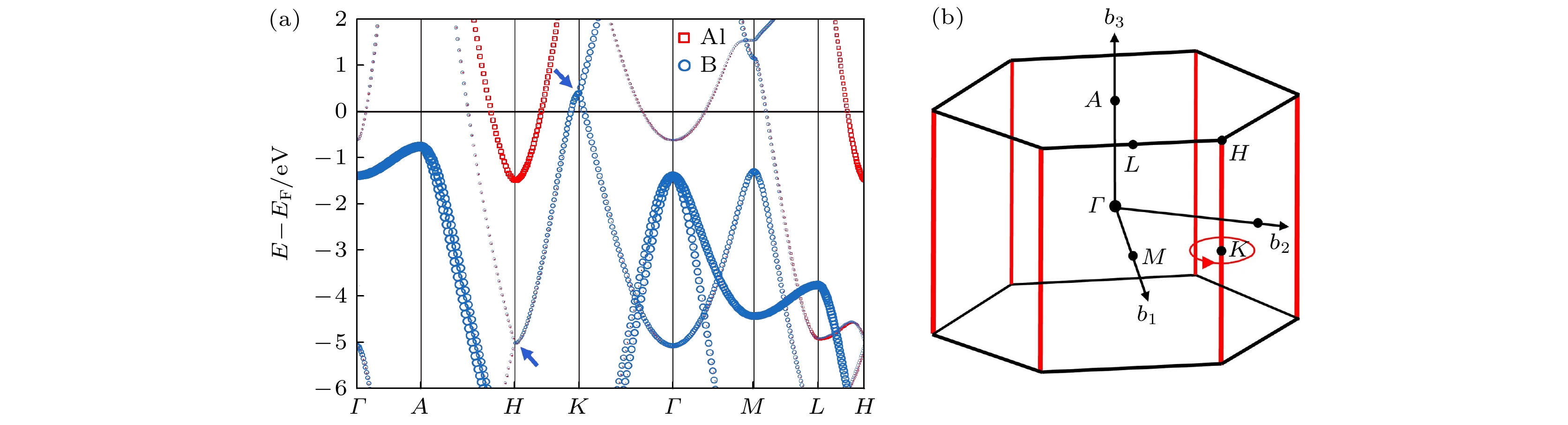
拓扑半金属, 作为一种独特的拓扑电子态, 近年来因其固有的拓扑特性和潜在的器件应用而备受研究关注. 体-边界对应是拓扑半金属的一个关键特征, 这意味着如果在体相中存在非平庸的能带拓扑, 那么在表面会存在受拓扑保护导电状态, 即拓扑表面态 (topological surface state, TSS). 这一特征使其在电催化研究领域备受关注. 本文采用第一性原理计算手段研究了AlB2材料的拓扑性质, 并构建(010)表面Al端面和B端面平板模型, 计算得到了TSS的位置. 研究了AlB2表面吸附特性, 发现Al端面氢吸附吉布斯自由能(
$ \Delta {G}_{{{\mathrm{H}}}^{*}} $ )仅为–0.031 eV, 相较于商业电催化析氢(hydrogen evolution reaction, HER)催化剂Pt的–0.08 eV更接近于0, 展现出了优异的HER性能. 观察了AlB2表面吸附H, OH和H2O后TSS的变化, 发现当H吸附时TSS变化最显著, 其次是OH吸附, H2O因其电中性和弱的表面吸附对TSS的影响很微弱. 并且在吸附前后, 由于受拓扑保护TSS依旧存在, 仅是能量发生了改变, 证实了其在环境中的稳定性. 本工作研究结果为不同吸附物对AlB2的TSS的影响提供了系统的认识, 为今后相关领域的理论和实验研究铺平了道路, 也为拓扑材料的实际应用提供了理论支撑.Topological semimetals have aroused great research interest due to their intrinsic topological physics and potential applications in devices. A key feature for all topological materials is the so-called bulk-boundary correspondence, which means that if there is non-trivial band topology in the bulk, then we can expect unique topologically protected conducting states in the surface, i.e. the topological surface state (TSS). Previously, the studies of the surface states of topological materials mainly focused on the pristine surfaces, while the topological nodal line semimetal surface states with adsorbates are rarely systematically studied. In this paper, the topological properties of the topological semimetal AlB2 are studied by first-principles calculations, and the TSS position is calculated by constructing the Al- and B-terminated slab models. Observing the topological surface state, it is found that the drumhead-like TSS connects two Dirac nodes with no energy gaps on the node line, and the TSS of the Al end-terminated slab has a smaller energy dispersion than that of the B-terminated slab. The adsorption characteristics of AlB2 (010) surface are studied, and it is found that the Gibbs free energy ($ {\Delta }{G}_{{{\mathrm{H}}}^{*}} $ ) for hydrogen adsorption on the surface of Al is only –0.031 eV, demonstrating excellent hydrogen evolution reaction (HER) performance. The changes of TSS after H, OH and H2O are adsorbed on the surface of AlB2 in aqueous solution environment are observed. The TSS change is the most significant when H is adsorbed, followed by OH adsorption. And the influence of H2O on TSS due to its electrical neutrality and weak surface adsorption is very weak. Before and after adsorption, because the topology protection TSS still exists, only the energy changes, which confirms its robustness in the environment. The results of this work provide a systematic understanding of the effects of different adsorbents on the TSS of AlB2, paving the way for future theoretical and experimental research in related fields, and alsopresent theoretical support for putting the topological materials into practical applications .-
Keywords:
- topological semimetal /
- topological surface state /
- surface adsorption
[1] Ando Y 2013 J. Phys. Soc. Jpn. 82 102001
 Google Scholar
Google Scholar
[2] Sato M, Ando Y 2017 Rep. Prog. Phys. 80 076501
 Google Scholar
Google Scholar
[3] Gao H, Venderbos J W F, Kim Y, Rappe A M 2019 Annu. Rev. Mater. Res. 49 153
 Google Scholar
Google Scholar
[4] Yan B, Felser C 2017 Annu. Rev. Conden. Ma. P. 8 337
 Google Scholar
Google Scholar
[5] Wang Z J, Sun Y, Chen X Q, Franchini C, Xu G, Weng H M, Dai X, Fang Z 2012 Phys. Rev. B 85 195320
 Google Scholar
Google Scholar
[6] Fang C, Weng H M, Dai X, Fang Z 2016 Chin. Phys. B 25 117106
 Google Scholar
Google Scholar
[7] Liu Z K, Zhou B, Zhang Y, Wang Z J, Weng H M, Prabhakaran D, Mo S K, Shen Z X, Fang Z, Dai X, Hussain Z, Chen Y L 2014 Science 343 864
 Google Scholar
Google Scholar
[8] Li G, Zhu S Y, Fan P, Cao L, Gao H J 2022 Chin. Phys. B 31 080301
 Google Scholar
Google Scholar
[9] Hasan M Z, Xu S Y, Belopolski I, Huang S M 2017 Annu. Rev. Conden. Ma. P. 8 289
 Google Scholar
Google Scholar
[10] Zhu Z M, Yu Z M, Wu W K, Zhang L F, Zhang W, Zhang F, Yang S A 2019 Phys. Rev. B 100 161401
 Google Scholar
Google Scholar
[11] Gao W S, Zhu M C, Chen D, Liang X, Wu Y L, Zhu A K, Han Y Y, Li L, Liu X, Zheng G L, Lu W J, Tian M L 2023 ACS Nano 17 4913
 Google Scholar
Google Scholar
[12] Wang L R, Yang Y, Wang J H, Liu W, Liu Y, Gong J L, Liu G D, Wang X T, Cheng Z X, Zhang X M 2022 EcoMat 5 e12316
 Google Scholar
Google Scholar
[13] Xie R K, Zhang T, Weng H M, Chai G L 2022 Small Sci. 2 2100106
 Google Scholar
Google Scholar
[14] Zhang G, Wu H, Zhang L, Yang L, Xie Y, Guo F, Li H, Tao B, Wang G, Zhang W, Chang H 2022 Small 18 2204380
 Google Scholar
Google Scholar
[15] Wang L, Zhao M, Wang J, Liu Y, Liu G, Wang X, Zhang G, Zhang X 2023 ACS Appl. Mater. Interfaces 15 51225
 Google Scholar
Google Scholar
[16] Chen H, Zhu W G, Xiao D, Zhang Z Y 2011 Phys. Rev. Lett. 107 056804
 Google Scholar
Google Scholar
[17] Li L Q, Zeng J, Qin W, Cui P, Zhang Z Y 2019 Nano Energy 58 40
 Google Scholar
Google Scholar
[18] Qu Q, Liu B, Liang J, Li H, Wang J N, Pan D, Sou I K 2020 ACS Catal. 10 2656
 Google Scholar
Google Scholar
[19] Li G W, Huang J, Yang Q, Zhang L G, Mu Q G, Sun Y, Parkin S, Chang K, Felser C 2021 J. Energy Chem. 62 516
 Google Scholar
Google Scholar
[20] Wei Y H, Ma D S, Yuan H K, Wang X, Kuang M Q 2023 Phys. Rev. B 107 235414
 Google Scholar
Google Scholar
[21] Rajamathi C R, Gupta U, Kumar N, Yang H, Sun Y, Süß V, Shekhar C, Schmidt M, Blumtritt H, Werner P, Yan B, Parkin S, Felser C, Rao C N R 2017 Adv. Mater. 29 1606202
 Google Scholar
Google Scholar
[22] Liu W, Zhang X M, Meng W Z, Liu Y, Dai X F, Liu G D 2022 iScience 25 103543
 Google Scholar
Google Scholar
[23] Kong X P, Jiang T, Gao J J, Shi X B, Shao J, Yuan Y H, Qiu H J, Zhao W W 2021 J. Phys. Chem. Lett. 12 3740
 Google Scholar
Google Scholar
[24] Li J X, Ma H, Xie Q, Feng S B, Ullah S, Li R H, Dong J H, Li D Z, Li Y Y, Chen X Q 2018 Sci. China Mater. 61 23
 Google Scholar
Google Scholar
[25] Jovic V, Consiglio A, Smith K E, Jozwiak C, Bostwick A, Rotenberg E, Di Sante D, Moser S 2021 ACS Catal. 11 1749
 Google Scholar
Google Scholar
[26] Wang L R, Zhang X M, Meng W Z, Liu Y, Dai X F, Liu G D 2021 J. Mater. Chem. A 9 22453
 Google Scholar
Google Scholar
[27] Ren Z B, Zhang H N, Wang S H, Huang B B, Dai Y, Wei W 2022 J. Mater. Chem. A 10 8568
 Google Scholar
Google Scholar
[28] Tang M Y, Shen H M, Qie Y, Xie H H, Sun Q 2019 J. Phys. Chem. C 123 2837
 Google Scholar
Google Scholar
[29] Kong X P, Shi X B, Zhao W W 2023 J. Phys. Chem. C 127 5271
 Google Scholar
Google Scholar
[30] Zhang X M, Wang L R, Li M H, et al. 2023 Mater. Today 67 23
 Google Scholar
Google Scholar
[31] Loa I, Kunc K, Syassen K, Bouvier P 2002 Phys. Rev. B 66 134101
 Google Scholar
Google Scholar
[32] Takane D, Souma S, Nakayama K, Nakamura T, Oinuma H, Hori K, Horiba K, Kumigashira H, Kimura N, Takahashi T, Sato T 2018 Phys. Rev. B 98 041105
 Google Scholar
Google Scholar
[33] Greeley J, Jaramillo T F, Bonde J, Chorkendorff I B, Norskov J K 2006 Nat. Mater. 5 909
 Google Scholar
Google Scholar
-
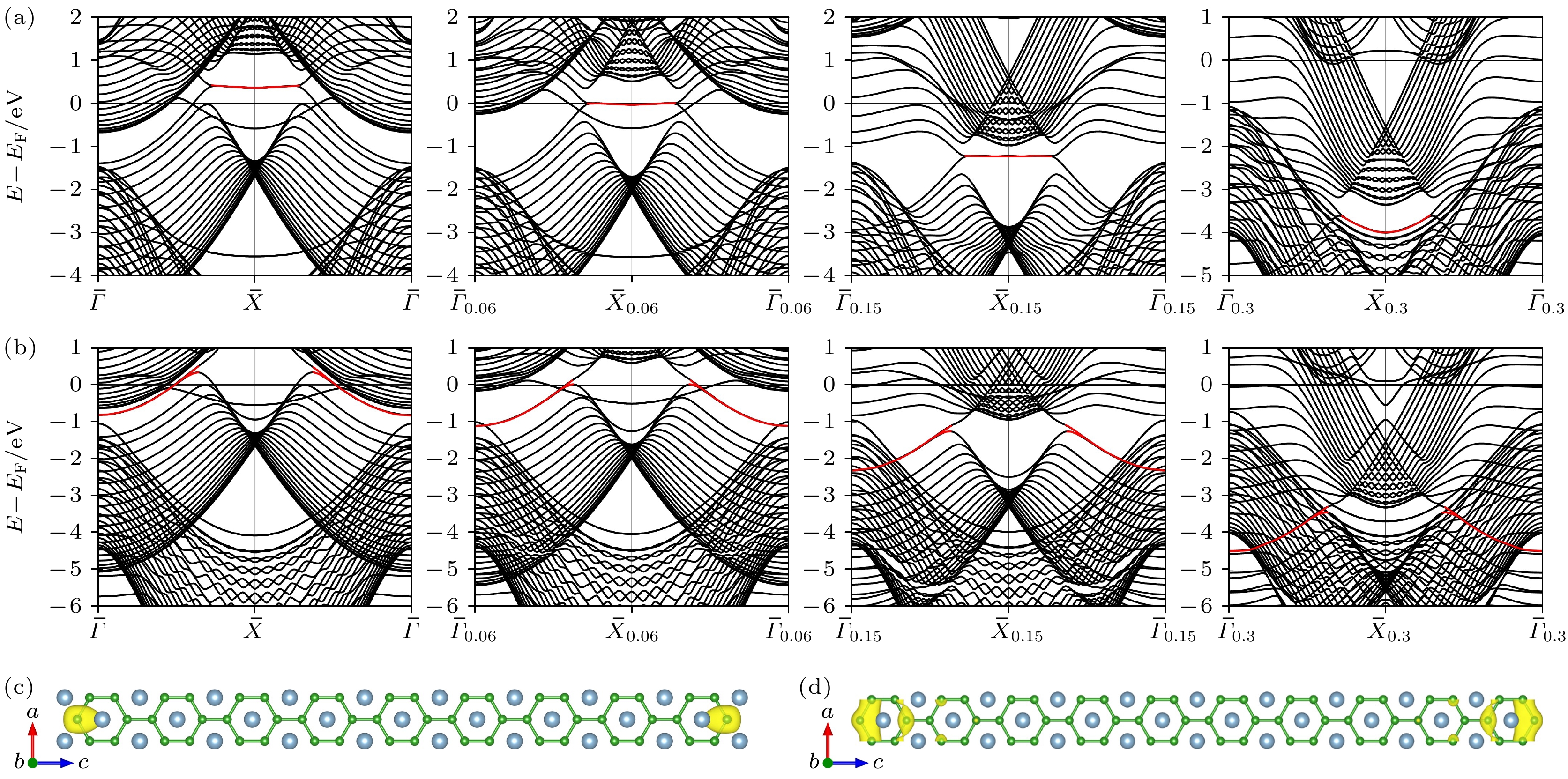
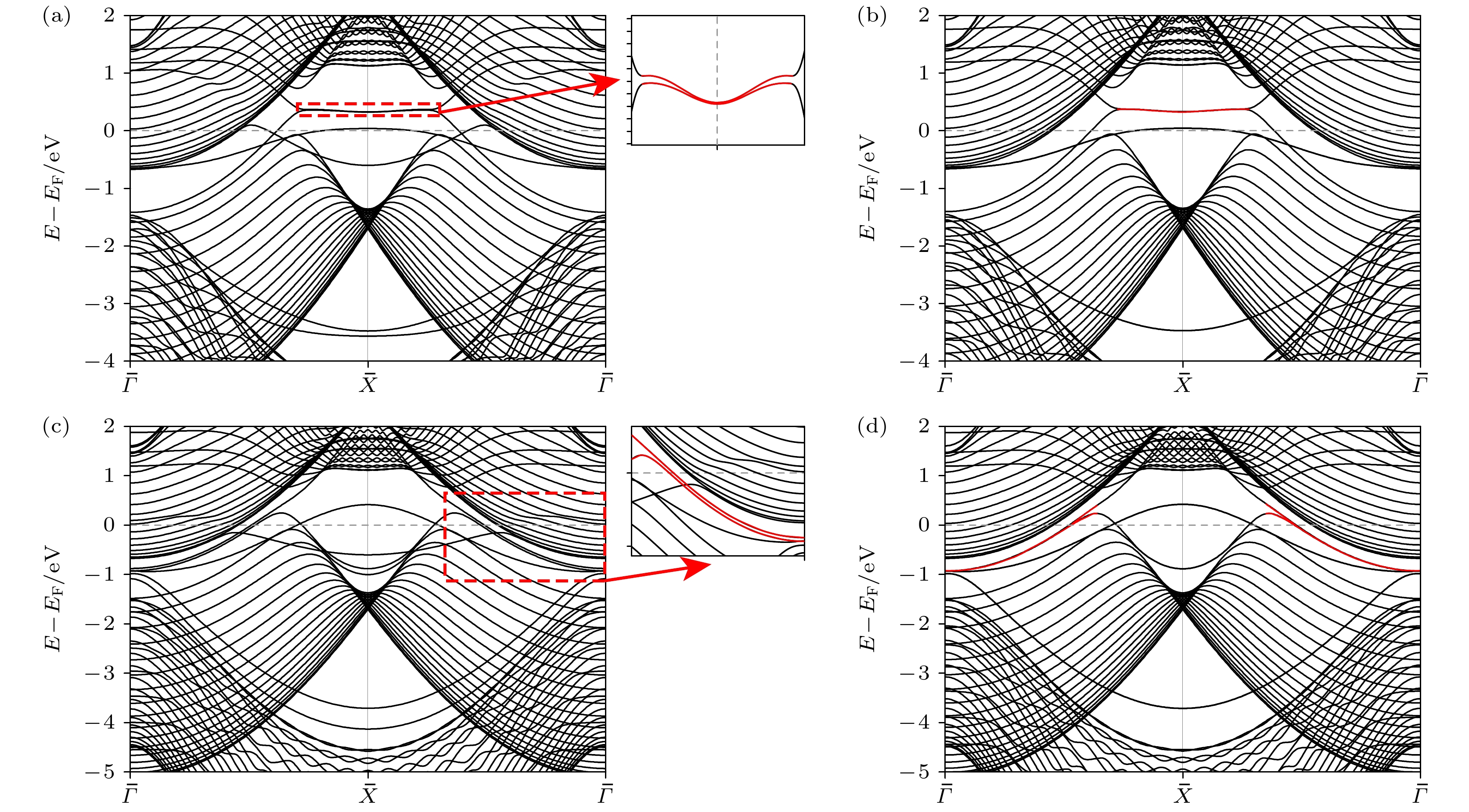
图 3 AlB2 (010) 20层平板的能带结构 (a)表面BZ中ksy = 0, 0.06, 0.15和0.3时, Al端面能带结构图; (b) ksy = 0, 0.1, 0.15和0.3处B端面能带结构图, 动量位置如图1(b)所示; (c) Al端面平板模型在图3(a)中红色标记的能带$ \bar{X} $高对称点处的部分电荷密度图; (d) B端面平板模型在图3(b)中红色标记的能带$ \bar{\varGamma } $高对称点处的部分电荷密度图
Fig. 3. Band structure of AlB2 (010) 20-layer slab: (a) Band structure of Al-terminated slab when ksy = 0, 0.06, 0.15 and 0.3 in surface BZ; (b) band structure of B-terminated slab at ksy = 0, 0.1, 0.15 and 0.3, and the position of high symmetry path, as shown in Fig. 1(b); (c) partial charge density of Al-terminated slab at the high symmetry point $ \bar{X} $ of the energy band marked in red in Fig. 3(a); (d) partial charge density diagram of B-terminated slab at the high symmetry point $ \bar{\varGamma } $ of the energy band marked in red in Fig. 3(b).
图 4 AlB2 (010)表面吸附最佳位点图 (a) Al端面top点位吸附H; (b) Al端面top点位吸附OH; (c) Al端面top点位吸附H2O; (d) B端面top点位吸附H; (e) B端面top点位吸附OH; (f) B端面top点位吸附H2O
Fig. 4. Optimal sites for the adsorption of AlB2 (010) surface: (a) Al-terminated slab adsorbing H on the top site; (b) Al-terminated slab adsorbing OH on the top site; (c) Al-terminated slab adsorbing H2O on the top site; (d) B-terminated slab adsorbing H on the top site; (e) B-terminated slab adsorbing OH on the top site; (f) B-terminated slab adsorbing H2O on the top site.
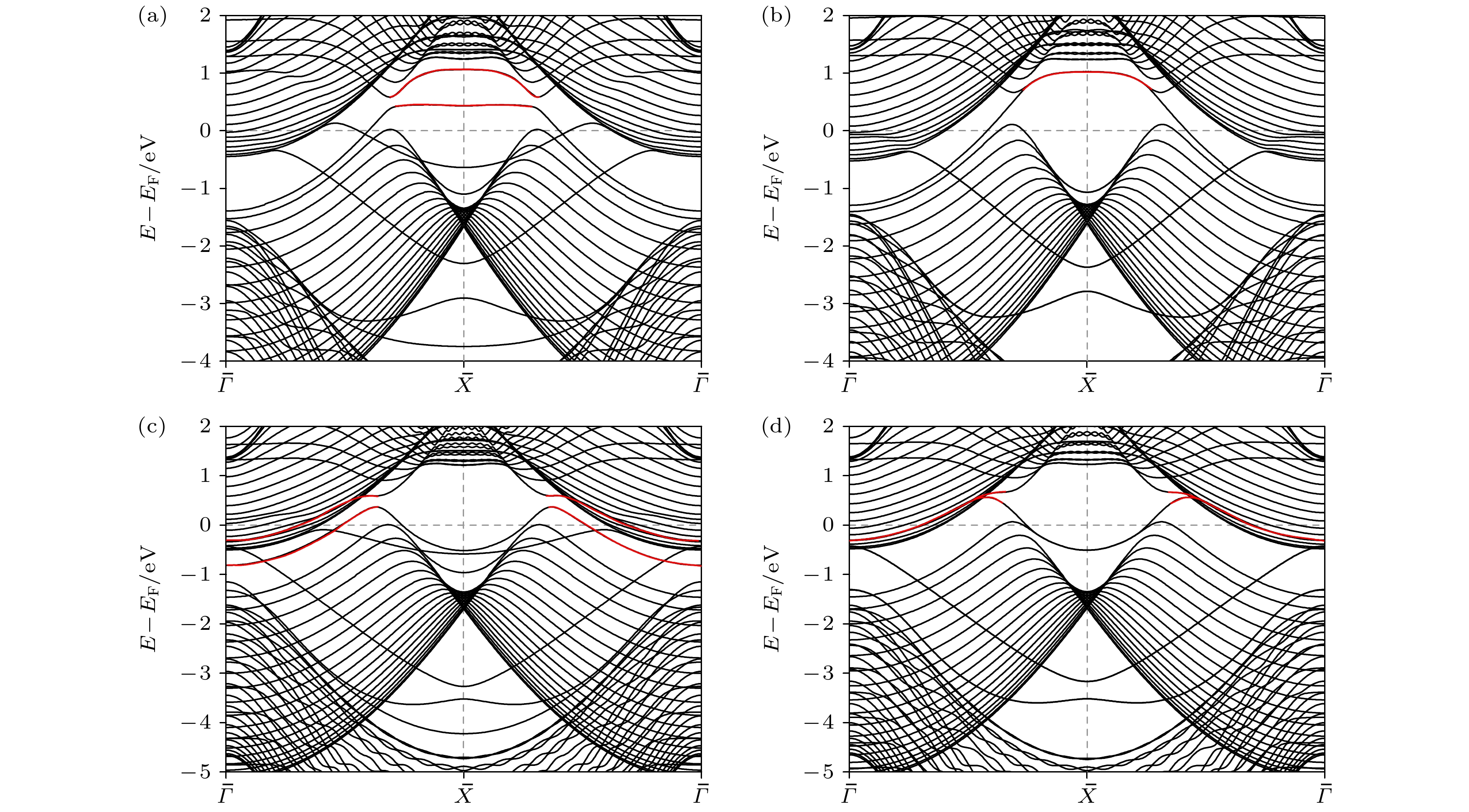
图 5 AlB2 (010) 表面TSS吸附H后的变化 (a) Al端面平板单侧吸附H; (b) Al端面平板两侧吸附H; (c) B端面平板单侧吸附H; (d) B端面平板两侧吸附H; 红色标记处为两节线间的鼓膜状TSS
Fig. 5. Changes of TSS adsorbing H of AlB2 (010) surface: (a) Al-terminated slab adsorbing H on a single side; (b) Al-terminated slab adsorbing H on both sides; (c) B-terminated slab adsorbing H on a single side; (d) B-terminated slab adsorbing H on both sides. The red line represents the drumhead-like TSS between two nodal lines.
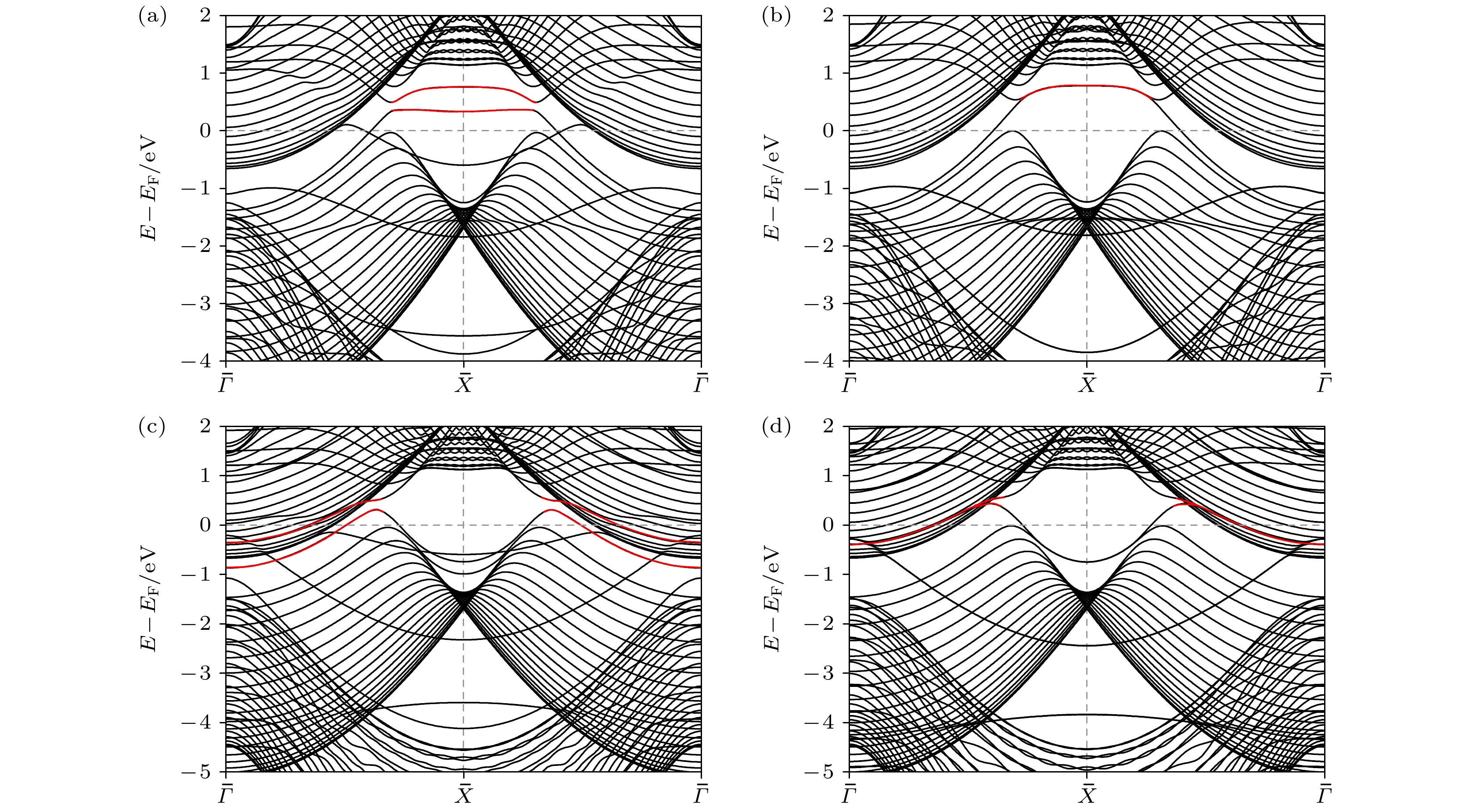
图 6 AlB2 TSS吸附OH后的变化 (a) Al端面平板单侧吸附OH; (b) Al端面平板两侧吸附OH; (c) B端面单侧吸附OH; (d) B端面平板两侧吸附OH; 红色标记处为两节线间的鼓膜状TSS
Fig. 6. Changes of TSS adsorbing OH of AlB2 (010) surface: (a) Al-terminated slab adsorbing OH on a single side; (b) Al-terminated slab adsorbing OH on both sides; (c) B-terminated slab adsorbing OH on a single side; (d) B-terminated slab adsorbing OH on both sides. The red line represents the drumhead-like TSS between two nodal lines.
图 7 AlB2 TSS吸附H2O后的变化 (a) Al端面平板单侧吸附H2O; (b) Al端面平板两侧吸附H2O; (c) B端面平板单侧吸附H2O; (d) B端面平板两侧吸附H2O; 红色标记处为两节线间的鼓膜状TSS
Fig. 7. Changes of TSS adsorbing H2O of AlB2 (010) surface: (a) Al-terminated slab adsorbing H2O on a single side; (b) Al-terminated slab adsorbing H2O on both sides; (c) B-terminated slab adsorbing H2O on a single side; (d) B-terminated slab adsorbing H2O on both sides. The red line represents the drumhead-like TSS between two nodal lines.
表 1 AlB2 (010) 表面吸附性能参数表, 对于Bader电荷分析“+”表示电荷的增益, 而“–”表示电荷的损失(单位为$ {{\mathrm{e}}}^{-} $)
Table 1. Parameters of adsorption properties of AlB2 (010) surface. For the Bader charge analysis, “+” means the gain of the charge, while “–” means the loss of the charge (unit in $ {{\mathrm{e}}}^{-} $).
材料 端面 吸附物 最佳位点 $ \Delta G/{\mathrm{eV}}$ Bader电荷分析/e– 吸附物 B Al AlB2 Al H top –0.031 +0.543 +0.073 –0.553 OH top –0.253 +0.737 +0.002 –0.751 H2O top 0.682 +0.368 +0.178 –0.556 B H top –1.659 +0.517 –0.174 –0.019 OH top –1.815 +0.717 –0.244 –0.008 H2O top 0.009 +0.384 –0.127 –0.001 -
[1] Ando Y 2013 J. Phys. Soc. Jpn. 82 102001
 Google Scholar
Google Scholar
[2] Sato M, Ando Y 2017 Rep. Prog. Phys. 80 076501
 Google Scholar
Google Scholar
[3] Gao H, Venderbos J W F, Kim Y, Rappe A M 2019 Annu. Rev. Mater. Res. 49 153
 Google Scholar
Google Scholar
[4] Yan B, Felser C 2017 Annu. Rev. Conden. Ma. P. 8 337
 Google Scholar
Google Scholar
[5] Wang Z J, Sun Y, Chen X Q, Franchini C, Xu G, Weng H M, Dai X, Fang Z 2012 Phys. Rev. B 85 195320
 Google Scholar
Google Scholar
[6] Fang C, Weng H M, Dai X, Fang Z 2016 Chin. Phys. B 25 117106
 Google Scholar
Google Scholar
[7] Liu Z K, Zhou B, Zhang Y, Wang Z J, Weng H M, Prabhakaran D, Mo S K, Shen Z X, Fang Z, Dai X, Hussain Z, Chen Y L 2014 Science 343 864
 Google Scholar
Google Scholar
[8] Li G, Zhu S Y, Fan P, Cao L, Gao H J 2022 Chin. Phys. B 31 080301
 Google Scholar
Google Scholar
[9] Hasan M Z, Xu S Y, Belopolski I, Huang S M 2017 Annu. Rev. Conden. Ma. P. 8 289
 Google Scholar
Google Scholar
[10] Zhu Z M, Yu Z M, Wu W K, Zhang L F, Zhang W, Zhang F, Yang S A 2019 Phys. Rev. B 100 161401
 Google Scholar
Google Scholar
[11] Gao W S, Zhu M C, Chen D, Liang X, Wu Y L, Zhu A K, Han Y Y, Li L, Liu X, Zheng G L, Lu W J, Tian M L 2023 ACS Nano 17 4913
 Google Scholar
Google Scholar
[12] Wang L R, Yang Y, Wang J H, Liu W, Liu Y, Gong J L, Liu G D, Wang X T, Cheng Z X, Zhang X M 2022 EcoMat 5 e12316
 Google Scholar
Google Scholar
[13] Xie R K, Zhang T, Weng H M, Chai G L 2022 Small Sci. 2 2100106
 Google Scholar
Google Scholar
[14] Zhang G, Wu H, Zhang L, Yang L, Xie Y, Guo F, Li H, Tao B, Wang G, Zhang W, Chang H 2022 Small 18 2204380
 Google Scholar
Google Scholar
[15] Wang L, Zhao M, Wang J, Liu Y, Liu G, Wang X, Zhang G, Zhang X 2023 ACS Appl. Mater. Interfaces 15 51225
 Google Scholar
Google Scholar
[16] Chen H, Zhu W G, Xiao D, Zhang Z Y 2011 Phys. Rev. Lett. 107 056804
 Google Scholar
Google Scholar
[17] Li L Q, Zeng J, Qin W, Cui P, Zhang Z Y 2019 Nano Energy 58 40
 Google Scholar
Google Scholar
[18] Qu Q, Liu B, Liang J, Li H, Wang J N, Pan D, Sou I K 2020 ACS Catal. 10 2656
 Google Scholar
Google Scholar
[19] Li G W, Huang J, Yang Q, Zhang L G, Mu Q G, Sun Y, Parkin S, Chang K, Felser C 2021 J. Energy Chem. 62 516
 Google Scholar
Google Scholar
[20] Wei Y H, Ma D S, Yuan H K, Wang X, Kuang M Q 2023 Phys. Rev. B 107 235414
 Google Scholar
Google Scholar
[21] Rajamathi C R, Gupta U, Kumar N, Yang H, Sun Y, Süß V, Shekhar C, Schmidt M, Blumtritt H, Werner P, Yan B, Parkin S, Felser C, Rao C N R 2017 Adv. Mater. 29 1606202
 Google Scholar
Google Scholar
[22] Liu W, Zhang X M, Meng W Z, Liu Y, Dai X F, Liu G D 2022 iScience 25 103543
 Google Scholar
Google Scholar
[23] Kong X P, Jiang T, Gao J J, Shi X B, Shao J, Yuan Y H, Qiu H J, Zhao W W 2021 J. Phys. Chem. Lett. 12 3740
 Google Scholar
Google Scholar
[24] Li J X, Ma H, Xie Q, Feng S B, Ullah S, Li R H, Dong J H, Li D Z, Li Y Y, Chen X Q 2018 Sci. China Mater. 61 23
 Google Scholar
Google Scholar
[25] Jovic V, Consiglio A, Smith K E, Jozwiak C, Bostwick A, Rotenberg E, Di Sante D, Moser S 2021 ACS Catal. 11 1749
 Google Scholar
Google Scholar
[26] Wang L R, Zhang X M, Meng W Z, Liu Y, Dai X F, Liu G D 2021 J. Mater. Chem. A 9 22453
 Google Scholar
Google Scholar
[27] Ren Z B, Zhang H N, Wang S H, Huang B B, Dai Y, Wei W 2022 J. Mater. Chem. A 10 8568
 Google Scholar
Google Scholar
[28] Tang M Y, Shen H M, Qie Y, Xie H H, Sun Q 2019 J. Phys. Chem. C 123 2837
 Google Scholar
Google Scholar
[29] Kong X P, Shi X B, Zhao W W 2023 J. Phys. Chem. C 127 5271
 Google Scholar
Google Scholar
[30] Zhang X M, Wang L R, Li M H, et al. 2023 Mater. Today 67 23
 Google Scholar
Google Scholar
[31] Loa I, Kunc K, Syassen K, Bouvier P 2002 Phys. Rev. B 66 134101
 Google Scholar
Google Scholar
[32] Takane D, Souma S, Nakayama K, Nakamura T, Oinuma H, Hori K, Horiba K, Kumigashira H, Kimura N, Takahashi T, Sato T 2018 Phys. Rev. B 98 041105
 Google Scholar
Google Scholar
[33] Greeley J, Jaramillo T F, Bonde J, Chorkendorff I B, Norskov J K 2006 Nat. Mater. 5 909
 Google Scholar
Google Scholar
计量
- 文章访问数: 4155
- PDF下载量: 170
- 被引次数: 0

















 下载:
下载: